Clear Sky Science · en
Spatiotemporal transcriptomic profiling reveals upregulation of glycolysis pathway genes before overt tauopathy in the PS19 mouse model
Why Energy-Hungry Brain Cells Matter
Alzheimer’s disease and related dementias are often described in terms of clumps and tangles of damaged proteins in the brain. But long before memory fails, brain cells are quietly struggling to meet their enormous energy demands. This study uses a sophisticated “map” of gene activity in a mouse model of tau-related dementia to show that energy pathways in specific memory circuits rev up early—well before classic tau tangles appear—offering new clues to how and where the disease process begins.
Looking Inside a Vulnerable Memory Circuit
The researchers focused on PS19 mice, which produce a human mutant form of tau protein and gradually develop brain changes resembling those in Alzheimer’s disease. They examined small, precisely chosen regions of the hippocampus (a key memory center) and nearby cortex at three ages: 2, 6 and 8 months, spanning early, mid and late disease stages. Using spatial transcriptomics, a technology that measures gene activity while preserving where each signal comes from in the tissue, they could compare how thousands of genes switched on or off in each region over time. This approach let them ask not just “what changes?” but “what changes where and when?” 
Early Energy Shift Before Visible Damage
A striking pattern emerged in the CA3 subregion of the hippocampus at just 2 months of age. At this point, tau tangles are not yet obvious there, and standard markers of synapses still look normal. Yet CA3 already shows hundreds of altered genes, far more than neighboring regions. Many of these genes relate to energy production, especially glycolysis—a core pathway that converts glucose into usable cellular fuel. One standout gene, Pgk1, which makes a key glycolytic enzyme, is among the most strongly increased. Every brain-expressed gene encoding a glycolysis enzyme is upregulated in CA3, suggesting that this circuit is boosting its energy machinery in response to rising levels of abnormal tau, even before structural damage is apparent.
From Metabolic Stress to Brain-Wide Inflammation
As the mice age to 6 and 8 months, tau pathology spreads and intensifies, and the pattern of gene activity broadens. By 8 months, all examined brain regions show changes in genes involved in ATP production, oxidative phosphorylation and mitochondrial function, indicating widespread metabolic stress. At the same time, there is strong activation of microglia and astrocytes, the brain’s resident immune and support cells. Gene signatures previously linked to disease-associated microglia and astrocytes in human Alzheimer’s tissue appear prominently in these mice, especially in hippocampal regions and parts of the cortex. Many of the genes most tightly correlated with the density of tau tangles belong to inflammatory and complement pathways, which are known to drive synapse loss and neurodegeneration. 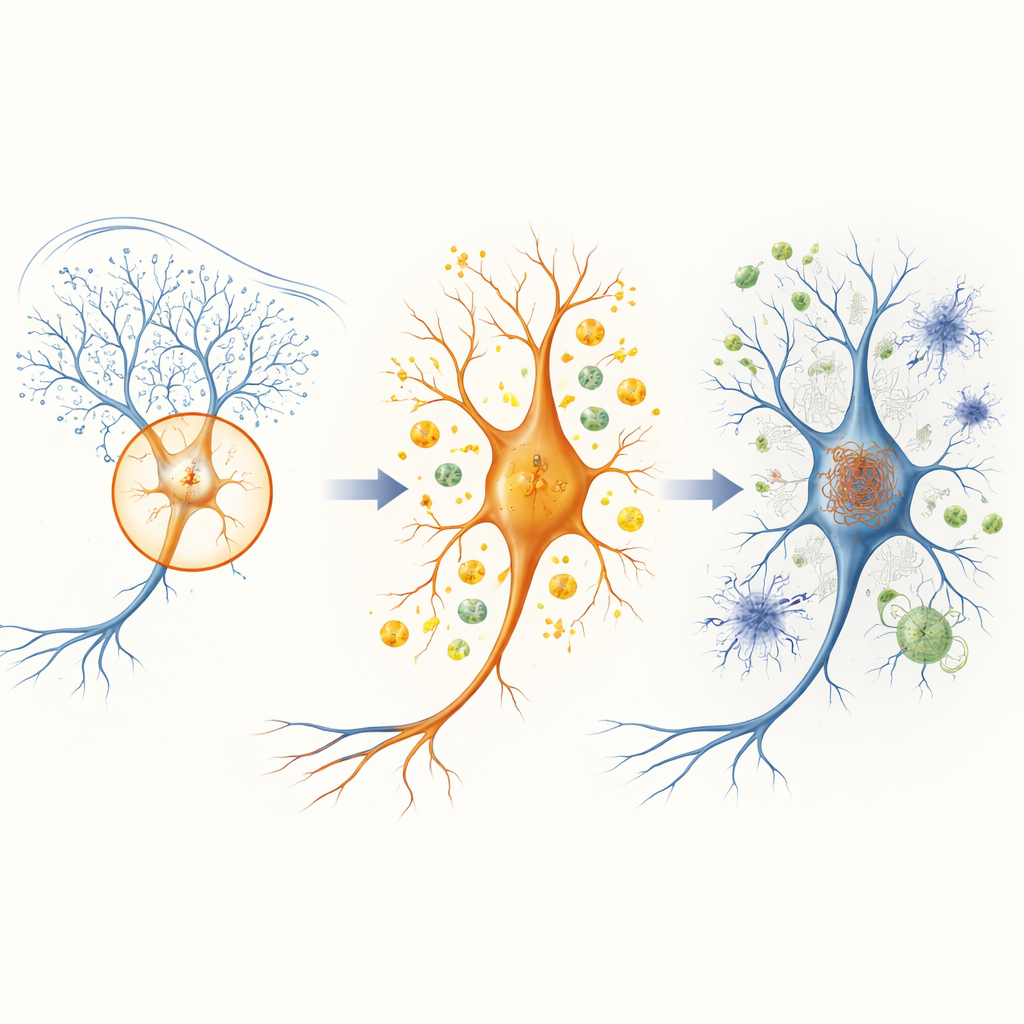
Timing and Location Shape Disease Progression
By tracking dynamic gene changes over time, the study reveals that hippocampal regions—particularly CA3 and the dentate gyrus—ramp up energy-related and protein-folding pathways early and then plateau. Cortical areas, in contrast, show a delayed but eventually similar shift in these same pathways between 6 and 8 months. This sequence mirrors the known spread of tau pathology from hippocampus to cortex. Across regions, the authors also identify shared signatures that hint at system-wide problems, including disrupted circadian rhythms and altered excitatory signaling, which align with sleep disturbances and memory decline reported in this model. Together, these spatial and temporal patterns argue that local cellular environments and regional vulnerabilities shape how tau pathology unfolds.
What This Means for Understanding and Treating Dementia
For a non-specialist, the key message is that in this tauopathy model, energy metabolism in a specific memory circuit goes into overdrive before visible tau tangles and major cell loss appear. The early boost in glycolysis, marked by Pgk1 and related genes, likely reflects an attempt by neurons to cope with rising stress from abnormal tau. Over time, this compensation seems to give way to chronic metabolic strain, widespread inflammation and eventual degeneration. By pinpointing when and where these changes occur, the work suggests that targeting metabolic pathways and glial activation in vulnerable regions—particularly the hippocampal CA3 area—at very early stages might slow or alter the trajectory of tau-driven diseases like Alzheimer’s.
Citation: Wang, S., Ponnusamy, M., Patel, O. et al. Spatiotemporal transcriptomic profiling reveals upregulation of glycolysis pathway genes before overt tauopathy in the PS19 mouse model. Exp Mol Med 58, 548–561 (2026). https://doi.org/10.1038/s12276-026-01652-z
Keywords: tauopathy, Alzheimer’s disease, hippocampus, brain metabolism, spatial transcriptomics