Clear Sky Science · en
Metabolic crosstalk among cancer-associated fibroblasts, adipocytes and immune cells as an immunosuppressive tumor microenvironment driver
Why the Tumor’s Neighborhood Matters
Cancer does not grow in isolation. It lives in a busy neighborhood of support cells, fat cells and immune cells that constantly trade nutrients and chemical messages. This article explains how that neighborhood’s “metabolism” – the way cells use and share fuel – quietly tilts the battle in favor of the tumor and against the immune system. Understanding this hidden economy of sugars, fats and amino acids is opening new ways to make immunotherapies work better and to cut off the tumor’s support system.
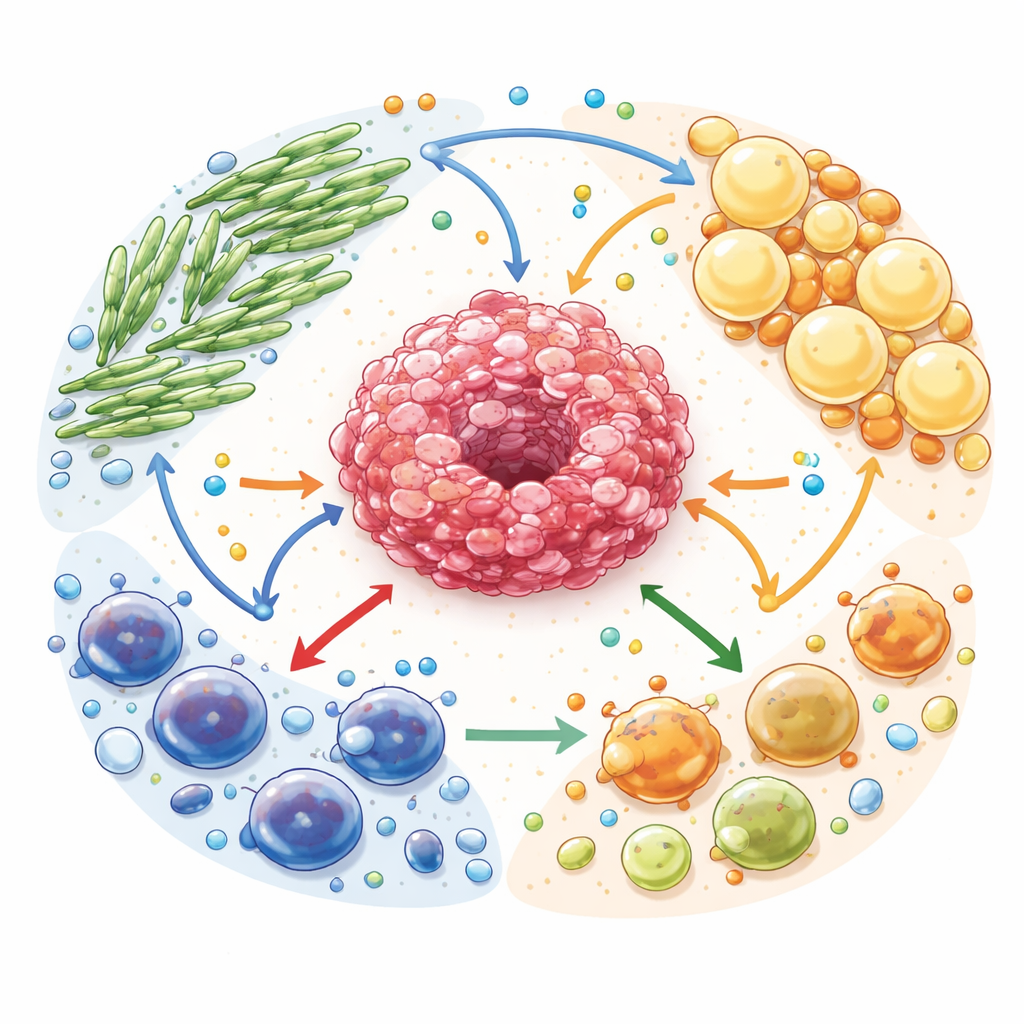
Fuel Sharing in a Cancer Ecosystem
The authors describe the tumor microenvironment as a complex ecosystem. Cancer cells are surrounded by cancer-associated fibroblasts (scar‑like support cells), cancer-associated adipocytes (reprogrammed fat cells) and many types of immune cells. All of these cells compete for and exchange fuels such as glucose, fatty acids and amino acids. But this trade is not neutral: support cells are rewired to break down their own stores and export energy-rich molecules that feed cancer cells. At the same time, waste-like byproducts, including lactate and adenosine, accumulate and act as powerful signals that weaken protective immune cells while favoring those that dampen immune attack.
How Fat Cells Help Tumors and Hinder Defenses
Nearby fat pads do far more than store calories. Under the influence of tumor signals and low oxygen, ordinary fat cells transform into cancer-associated adipocytes. They shrink their fat droplets, ramp up fat breakdown and pour out free fatty acids, inflammatory molecules and tiny cargo-filled vesicles. Cancer cells greedily import these fats and burn them in their mitochondria, gaining a flexible, long‑lasting source of energy that helps them survive stress, spread and resist treatment. Immune cells in fat‑rich regions do not fare as well: killer T cells and natural killer cells overload on fats, suffer from oxidative stress, and slide into an “exhausted” state with blunted ability to destroy tumor cells. In contrast, regulatory T cells and certain myeloid cells thrive on these lipids, becoming more suppressive and further muffling immune responses.
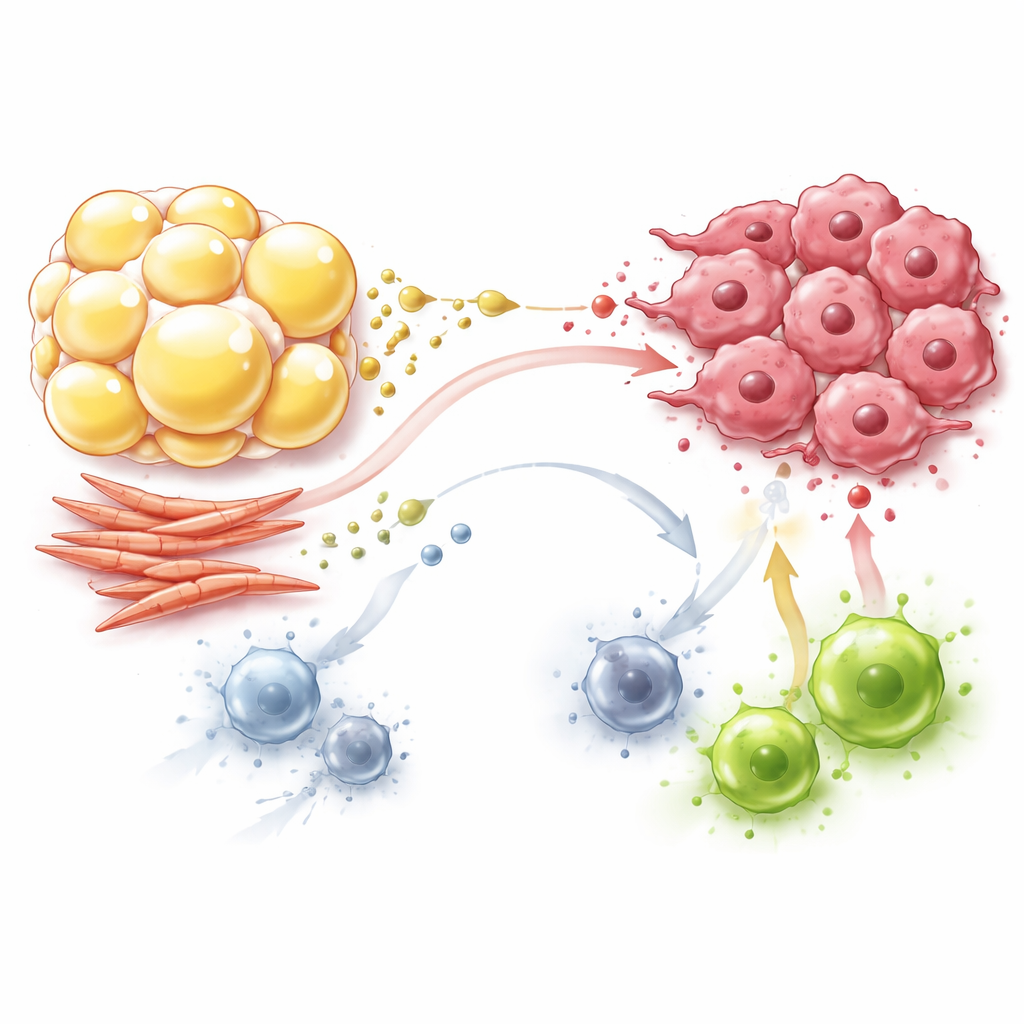
Fibroblasts: Builders That Also Rewire Metabolism
Fibroblasts, long known as the builders of scar‑like tissue around tumors, are also active players in this metabolic drama. Cancer-associated fibroblasts shift toward sugar‑hungry behavior, turning large amounts of glucose into lactate and pyruvate even when oxygen is available. They export these products into the surrounding space, where cancer cells reuse them as fuel for their own power plants, sparing glucose for growth. Fibroblasts also manufacture and trade amino acids such as glutamine, proline and asparagine, which support both the tumor’s structure and its growth under nutrient stress. At the same time, fibroblasts consume key nutrients and release lactate, crowding out effector T cells that depend on glucose and pushing macrophages and T cells toward more tolerant, less aggressive states.
Immune Cells Caught in a Metabolic Trap
The immune system inside tumors is shaped as much by fuel availability as by antigens. Cytotoxic T cells and natural killer cells, which need rapid sugar burning to power their attack, find themselves in a landscape stripped of glucose and flooded with lactate, fats and other suppressive metabolites. Their engines stall, their mitochondria are damaged and inhibitory “brakes” on their surfaces increase. Meanwhile, regulatory T cells and tumor‑associated macrophages are well adapted to this harsh environment. They favor fat burning and oxidative metabolism, allowing them to prosper where others fail. Supported by fibroblasts and adipocytes, these cells reinforce a tolerant, wound‑healing‑like state that protects the tumor and blunts the impact of checkpoint inhibitors and other immunotherapies.
Turning Metabolism into a Therapeutic Target
The review highlights a growing toolbox of experimental drugs designed to disrupt this support network without simply poisoning dividing cells. Some approaches aim to block fat release from adipocytes or fat uptake and burning in cancer cells. Others target fibroblast‑driven pathways that generate lactate, remodel the matrix or attract suppressive myeloid cells. By lowering lactate levels, blocking specific fuel transporters or reprogramming lipid‑sensing switches inside cells, these strategies seek to restore glucose access and mitochondrial health to exhausted T and natural killer cells. The ultimate goal is not only to starve the tumor, but to “re‑educate” its neighborhood so that immune cells once again see, reach and effectively attack cancer cells.
Citation: Kim, T.H., Lim, S.H., Lee, H. et al. Metabolic crosstalk among cancer-associated fibroblasts, adipocytes and immune cells as an immunosuppressive tumor microenvironment driver. Exp Mol Med 58, 366–381 (2026). https://doi.org/10.1038/s12276-026-01650-1
Keywords: tumor microenvironment, cancer metabolism, cancer-associated fibroblasts, cancer-associated adipocytes, tumor immunosuppression