Clear Sky Science · en
Supplementation of L-aspartate corrects MASLD and MASH in mice by inhibiting platelet–hepatocyte interaction-mediated mitochondrial fragmentation via the ATP–P2X7–NEK7–DRP1 axis
Why this liver study matters
Many people carry extra fat in their livers without knowing it. This silent problem, now called metabolic dysfunction–associated steatotic liver disease (MASLD), can progress to serious inflammation, scarring and even liver cancer. The paper summarized here explores whether a simple natural amino acid, L-aspartate, can protect the liver in mice and uncovers an unexpected culprit in liver damage: overactive blood platelets that injure liver cell “power plants,” the mitochondria.
A common liver problem in modern life
MASLD is tightly linked to obesity and type 2 diabetes and may soon affect more than a third of the global population. In MASLD, liver cells swell with fat, become stressed and start to die, which triggers inflammation and scar formation. Healthy mitochondria normally burn fats and sugars for energy, but in this disease they become damaged and fragmented, so the liver burns less fat and accumulates more of it. Despite the scale of the problem, only one drug has been approved so far and it helps only a fraction of patients, so researchers are searching for safe, affordable treatments that restore the liver’s energy machinery.
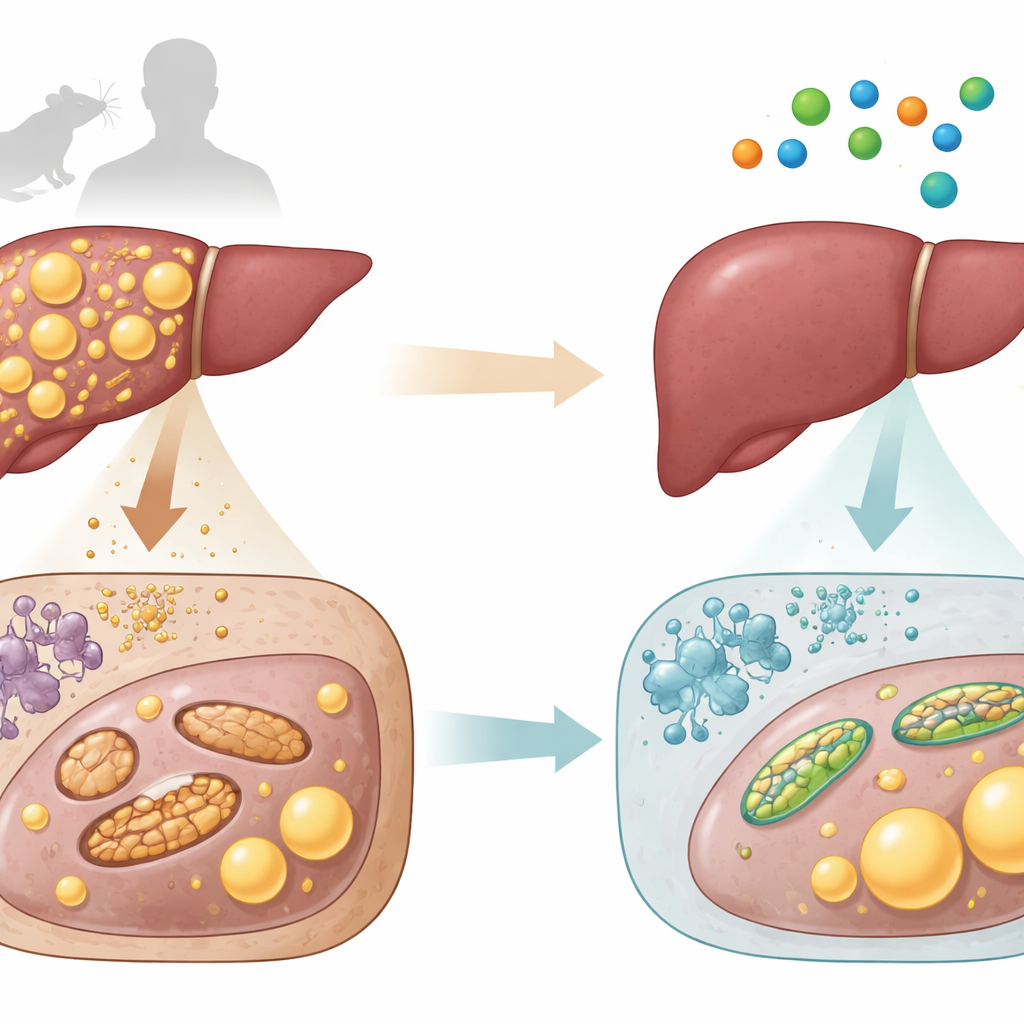
A simple amino acid as a potential helper
L-aspartate is a small building block of proteins that also participates in the body’s energy cycles and is already used clinically as a liver-supportive compound. The researchers first measured L-aspartate in blood and liver samples from mice and from people. They found that animals and human patients with fattier, more damaged livers had consistently lower levels of L-aspartate in their liver tissue. In both species, the less L-aspartate present, the higher the fat content and the stronger the signals of liver injury. This pattern suggested that falling L-aspartate might go hand-in-hand with worsening liver health.
Reversing fatty liver and inflammation in mice
The team then asked whether adding L-aspartate back could actually repair disease. They fed mice a high-fat, high-cholesterol diet or a special diet that provokes severe liver inflammation and scarring, mimicking human MASLD and its advanced form, MASH. When these mice received L-aspartate injections at doses comparable to those used in the clinic, their livers became lighter and less greasy, liver enzymes in the blood dropped, and microscopic exams showed fewer fat droplets, less inflammation and reduced scarring. These benefits appeared in both early fatty liver and advanced inflammatory disease and were comparable to those of established reference drugs in the same models.
Protecting the cell’s power plants
Looking inside the liver, the scientists observed that diseased mice had fewer mitochondria, many of them enlarged and damaged or broken into tiny fragments. L-aspartate treatment restored a healthier network of elongated mitochondria. It boosted the activity of key energy-producing enzyme complexes, raised mitochondrial oxygen use and ATP output, and increased markers of mitochondrial “cleanup” and renewal. Whole-animal measurements showed higher oxygen consumption and energy expenditure, indicating that the mice were burning more fuel overall rather than simply eating less.
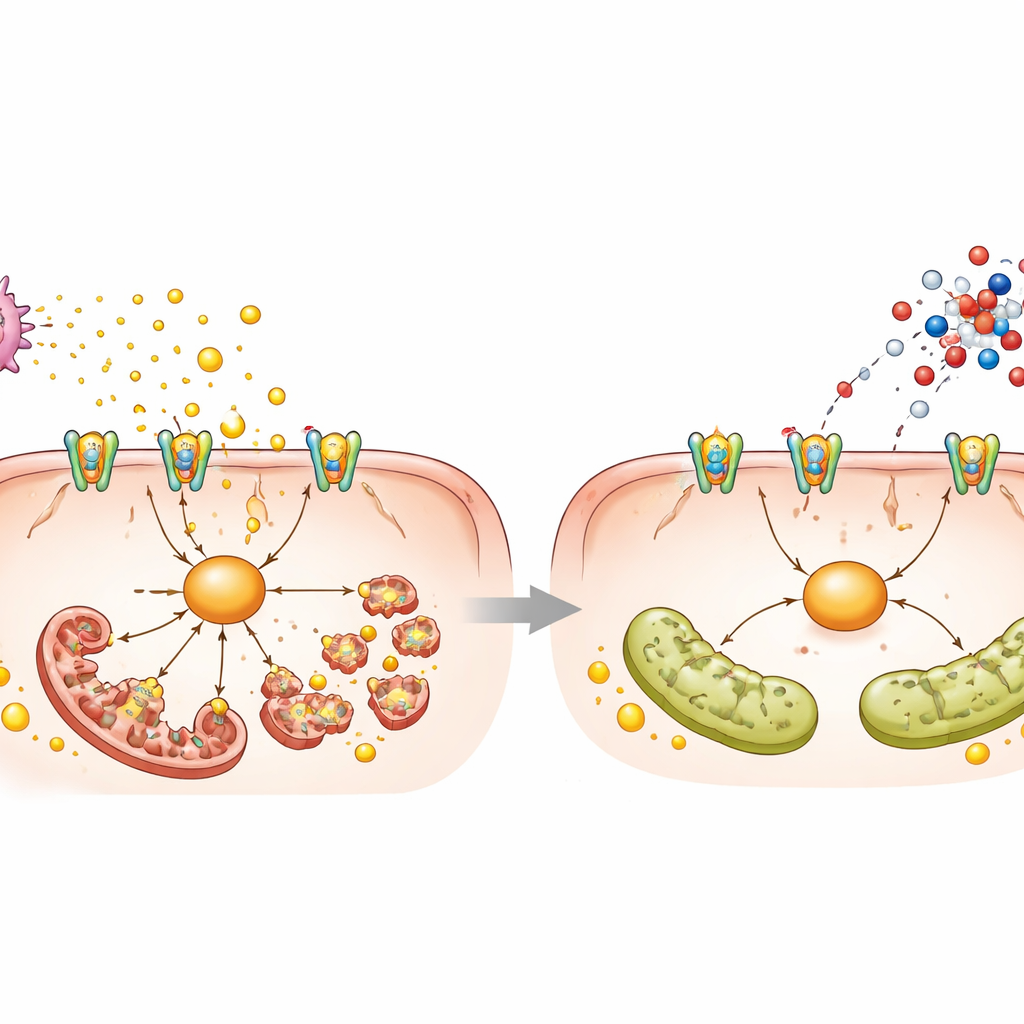
Platelets, a toxic signal, and a molecular chain
To understand how L-aspartate exerts these effects, the researchers combined gene activity and metabolite profiling after a single dose. An unexpected signal emerged: pathways linked to platelet activation and a messenger molecule called cGMP. In fatty liver, platelets in blood and liver were more numerous and activated, and they released extra ATP, a normal energy molecule that becomes harmful when dumped outside cells. This ATP activated a receptor called P2X7 on liver cells, which in turn switched on a protein called NEK7 and the fission protein DRP1, driving mitochondria to fragment and promoting fat buildup and cell death. L-aspartate raised cGMP levels inside platelets, dampened their activation and aggregation, reduced ATP release and calmed this P2X7–NEK7–DRP1 chain inside liver cells. Directly blocking platelets with aspirin, inhibiting P2X7, or knocking down NEK7 mimicked many of L-aspartate’s protective effects, supporting the idea that platelet–liver crosstalk is central to the damage.
What this could mean for patients
Together, these findings suggest that in fatty liver disease, overactive platelets shower the liver with ATP, which triggers a molecular cascade that shatters mitochondria and worsens fat accumulation and injury. In mice, supplementing L-aspartate breaks this loop: it keeps platelets calmer, limits ATP release, preserves mitochondrial shape and function, and reverses both simple fatty liver and inflammatory, fibrotic disease. Because L-aspartate is inexpensive and already used as a liver-supportive agent, the work highlights it as a promising candidate to test in human MASLD and MASH, while also pointing to platelet-driven mitochondrial damage as a new target for future therapies.
Citation: Cao, WJ., Su, R., Fu, HL. et al. Supplementation of L-aspartate corrects MASLD and MASH in mice by inhibiting platelet–hepatocyte interaction-mediated mitochondrial fragmentation via the ATP–P2X7–NEK7–DRP1 axis. Exp Mol Med 58, 533–547 (2026). https://doi.org/10.1038/s12276-026-01648-9
Keywords: fatty liver disease, platelets, mitochondria, L-aspartate, metabolic liver disease