Clear Sky Science · en
Microglia-associated progression of multiple sclerosis: target identification and therapeutic engagement in human in vitro models
Why Brain Immune Cells Matter in Multiple Sclerosis
Multiple sclerosis (MS) is often described as a disease of the body’s immune system attacking the brain and spinal cord. Many current drugs successfully reduce sudden attacks, or relapses, but they do not stop the slow, silent worsening that many patients experience over years. This review explains how tiny immune cells that live permanently in the brain, called microglia, may drive that long-term decline—and how new lab-grown human brain models are helping scientists find treatments that can finally slow or halt progression.
The Hidden Engine of Ongoing Damage
MS has traditionally been divided into relapsing and progressive forms, but evidence now shows that progression begins very early in almost everyone with the disease, even when they still appear to have only relapses. While relapses are fueled by immune cells entering from the blood, progression seems to be powered by inflammation trapped inside the brain and spinal cord. In this sealed-off environment, microglia are involved in many harmful processes: ongoing inflammation, loss of the insulating myelin coating on nerve fibers in both white and gray matter, buildup of damaging oxygen-related chemicals, and failure of repair. The end result is the gradual loss of nerve cells and their connections. Microglia normally help keep the brain healthy by clearing debris and supporting nerve cells, but in MS they often shift toward more aggressive states and lose some of their protective roles, making them prime suspects in driving progression.
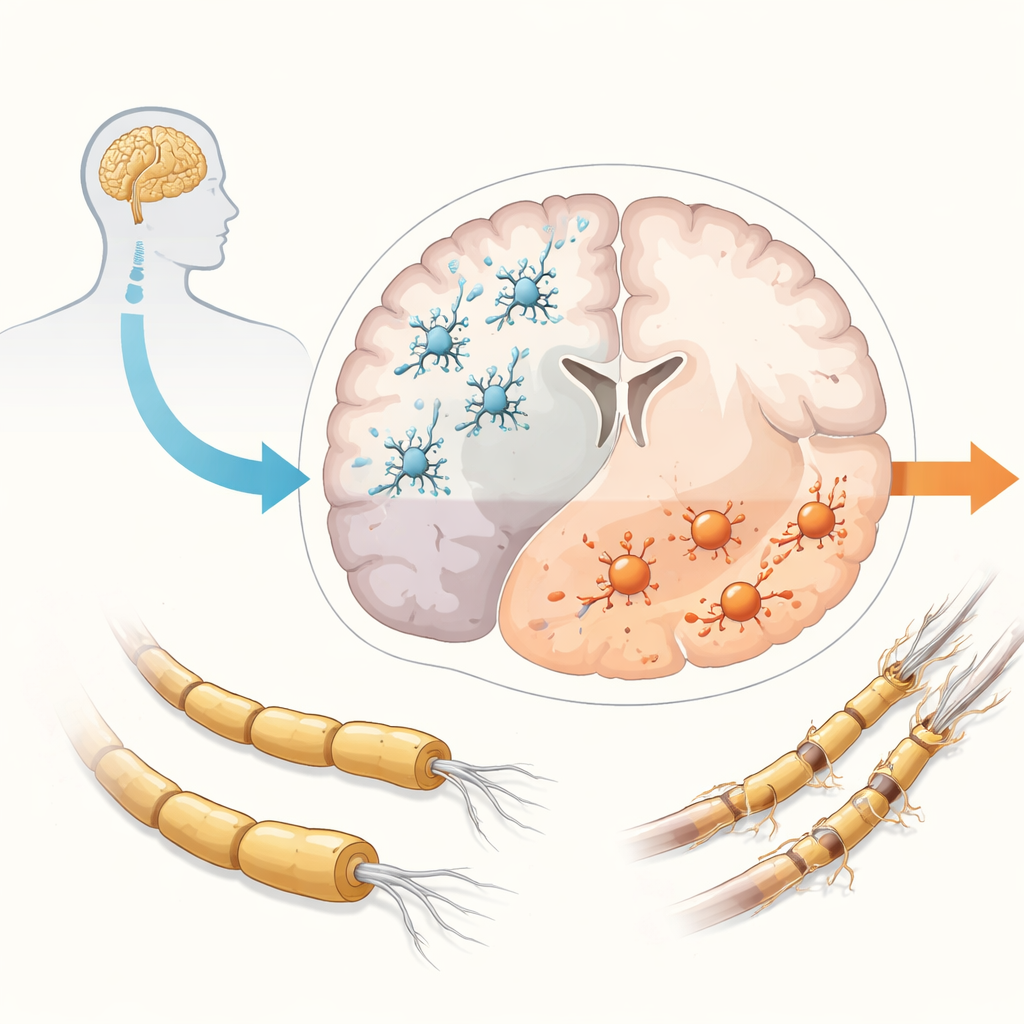
Why Traditional Animal Models Fall Short
For decades, studies in mice and other animals have been crucial for developing drugs that limit relapses, yet they have largely failed to yield therapies that stop progression. One reason is that animal models only partially mimic human MS and do not fully reproduce the complex, long-lasting inflammation seen in patients’ brains. Microglia from rodents also differ from human microglia in important genes and responses. As a result, treatments that look promising in animals often do not work in people. The shortage of animal models that truly capture progressive MS has pushed researchers to build new human-based systems in the lab, where they can study microglia and other brain cells more directly.
Building Human Brain Models in a Dish
Scientists now use several layers of laboratory models to study human microglia. Primary cells taken directly from rodent or human brain tissue preserve many natural features but are hard to obtain, change quickly outside their native environment, and cannot easily be scaled up. To overcome these hurdles, researchers turn to induced pluripotent stem (iPS) cells—adult cells reprogrammed back into a flexible, stem-like state. These iPS cells can be guided to become microglia, neurons, or other brain cells. In simple flat cultures, iPS-derived microglia capture many key traits and can be produced in large numbers, enabling detailed experiments and drug screens. They can even be generated from individual patients with MS, revealing built-in differences such as altered gene activity and changes in how these cells respond to stress and clear debris.
Adding Realistic Brain Neighborhoods
Because microglia behavior is strongly shaped by their surroundings, researchers have moved beyond single-cell layers to more realistic setups. In two-dimensional cocultures, microglia grow together with neurons and supporting cells, which helps them adopt more natural shapes and behaviors and lets scientists probe how cell types influence one another. Three-dimensional spheroids and organoids—tiny, self-organizing pieces of brain-like tissue—go further, providing a soft, crowded environment that more closely resembles actual brain tissue. When microglia are embedded in these structures, they show complex branching shapes, react to injury, interact with a blood–brain barrier–like layer, and respond to inflammatory signals from the spinal fluid of people with MS. Such models have been used to study how chronic inflammation triggers a “tired yet inflamed” state called cellular senescence in microglia and astrocytes, and how changes in microglial fat handling may impair myelin repair. In parallel, transplanting human iPS-derived microglia or organoids into mouse brains allows these cells to mature further within a living network and to be tested in established MS-like disease models.
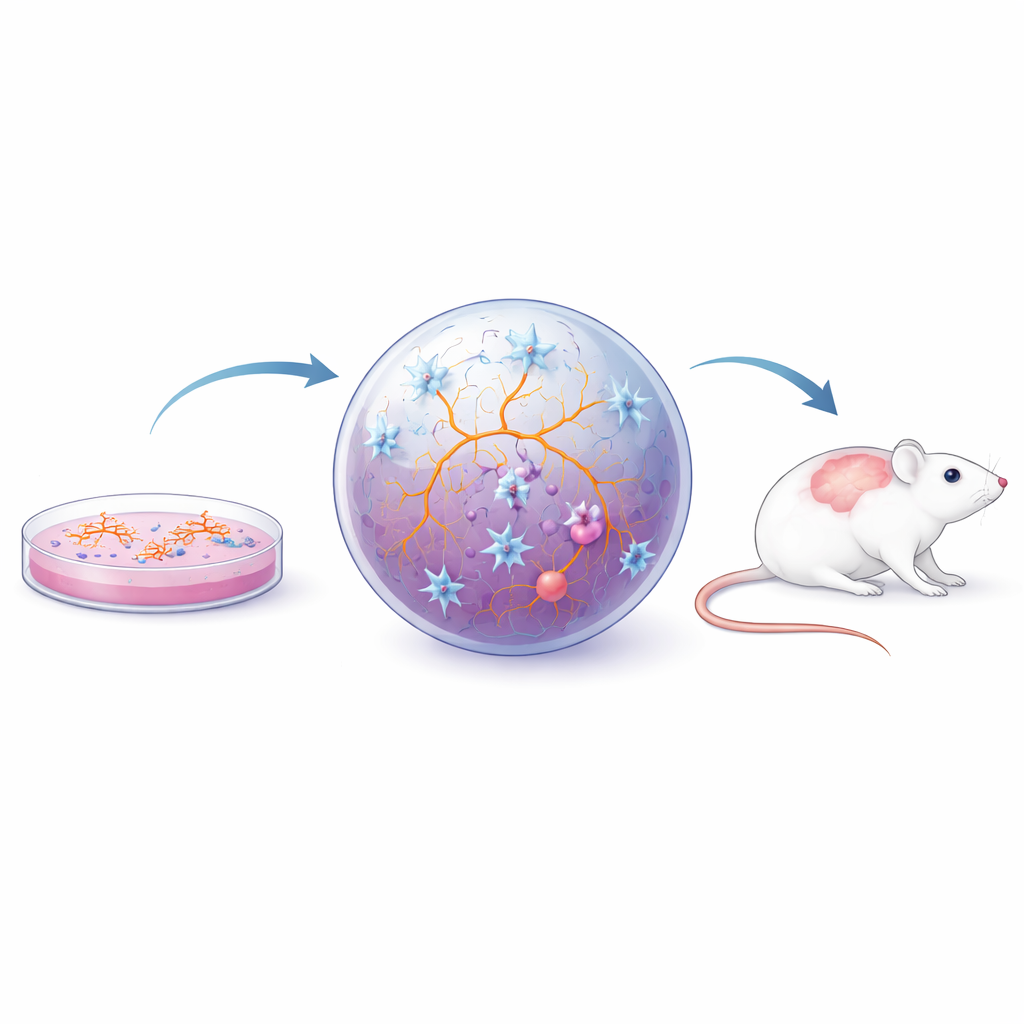
From Lab Models to Future Treatments
Taken together, these human-based in vitro and chimeric models form a toolkit for dissecting how microglia contribute to MS progression and for testing new therapies that aim to calm harmful microglial activity while restoring their protective functions. Although none of the models fully reproduces the human brain, each captures different pieces of the puzzle, and together they bridge the gap between simple cell cultures and imperfect animal models. By combining insights from these systems, researchers hope to identify precise drug targets—such as pathways controlling inflammation, aging-like changes, and lipid metabolism in microglia—that could finally translate into treatments that slow or stop the gradual disability that characterizes progressive MS.
Citation: Blenkle, A., Geladaris, A. & Weber, M.S. Microglia-associated progression of multiple sclerosis: target identification and therapeutic engagement in human in vitro models. Exp Mol Med 58, 357–365 (2026). https://doi.org/10.1038/s12276-026-01647-w
Keywords: multiple sclerosis progression, microglia, induced pluripotent stem cells, brain organoids, neuroinflammation