Clear Sky Science · en
A ESRP1/circPHGDH/miR-149/RAP1B positive feedback loop promotes the malignant behaviors and glycolysis of prostate cancer cell
Why this research matters for men's health
Prostate cancer is one of the most common cancers in men, and many tumors eventually become aggressive and resistant to treatment. This study uncovers a hidden control circuit inside prostate cancer cells that helps them grow faster, spread more easily, and rewire how they burn sugar for energy. By mapping this molecular loop, the researchers point to new weak spots that might be targeted to slow the disease or make existing therapies more effective.
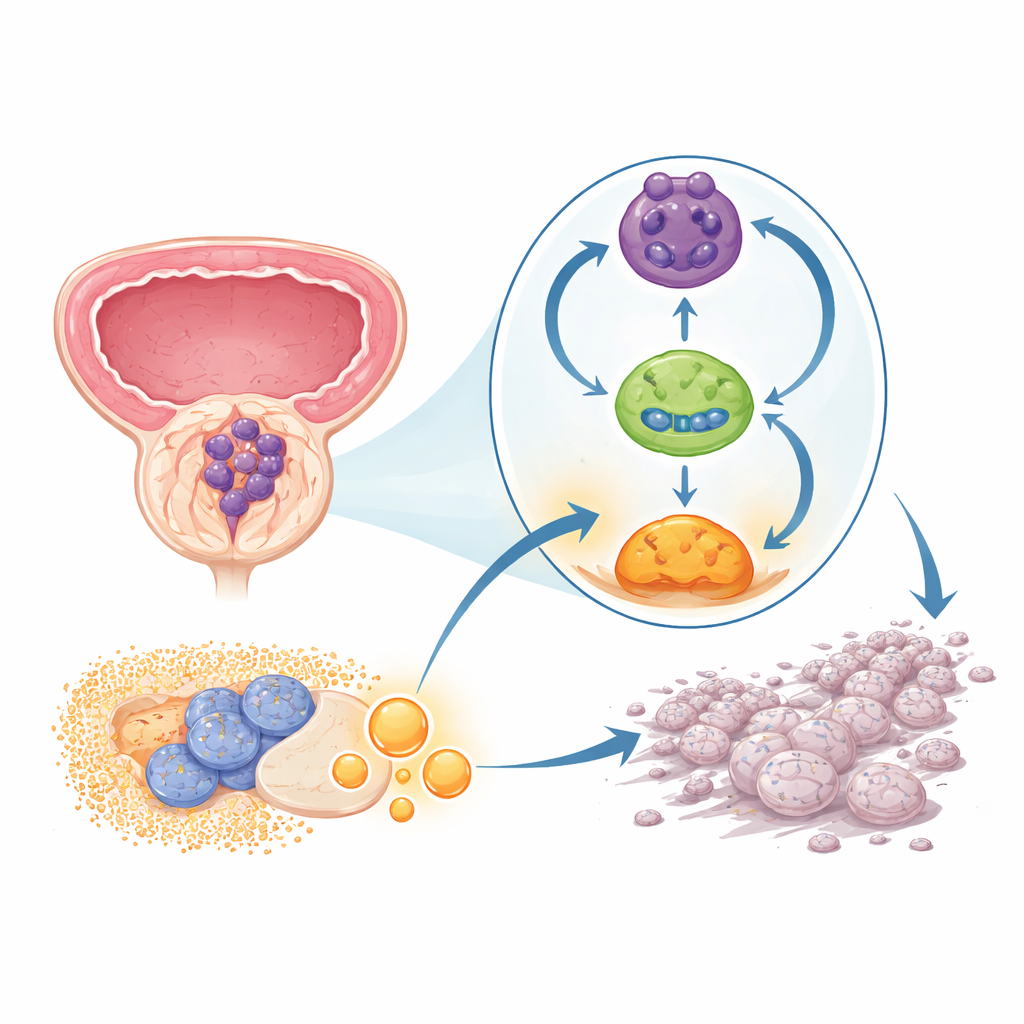
A hidden loop that keeps cancer switched on
The team focused on a newly appreciated class of genetic material called circular RNAs. Unlike the usual linear strands, these molecules form closed rings and are remarkably stable. In prostate tumor samples and cell lines, the researchers found one particular circular RNA, called circPHGDH, was consistently higher than in nearby noncancerous tissue. Patients whose tumors had more of this RNA tended to have larger tumors, more advanced local growth, and distant spread, hinting that circPHGDH helps drive a more dangerous form of the disease.
How cancer cells change their behavior
To see what circPHGDH actually does, the scientists altered its levels in prostate cancer cells grown in the lab. When they reduced circPHGDH, the cells formed fewer colonies, moved and invaded through membranes less efficiently, and showed signs of staying in a more "epithelial" state that is less prone to spreading. At the same time, the cells shifted away from a sugar-hungry metabolic style typical of many tumors: their reliance on glycolysis dropped, and they used oxygen-based energy production more. When circPHGDH was boosted instead, all of these cancer-promoting traits moved in the opposite direction.
Small RNAs and a growth signal relay
The study then traced how circPHGDH exerts these effects. In the cell’s watery interior, circPHGDH acts like a sponge for a tiny regulatory RNA called miR-149, soaking it up and preventing it from restraining its usual targets. One key target is RAP1B, a signaling protein that feeds into a major growth and survival pathway known to be important in prostate cancer. When miR-149 is held back by circPHGDH, RAP1B levels rise and switch on downstream signals that promote cell division, movement, and a glycolysis-heavy metabolism. Restoring miR-149 or directly reducing RAP1B reversed many of the harmful effects, both in cell cultures and in mice bearing human prostate tumors.
Metabolic waste that feeds the fire
Another layer of the story involves how circPHGDH is produced in the first place. Its creation depends on a splicing protein called ESRP1, which helps decide how raw RNA messages are cut and rejoined. The researchers showed that ESRP1 binds specific sites around the circPHGDH region and favors its circular form over the standard linear version. Crucially, they discovered that lactate—the end product of glycolysis—chemically modifies ESRP1 at a single site, making the protein more stable. Because circPHGDH itself pushes cells toward more glycolysis and thus more lactate, this creates a self-reinforcing loop: ESRP1 increases circPHGDH, circPHGDH boosts RAP1B and glycolysis, glycolysis generates lactate, and lactate, in turn, stabilizes ESRP1.
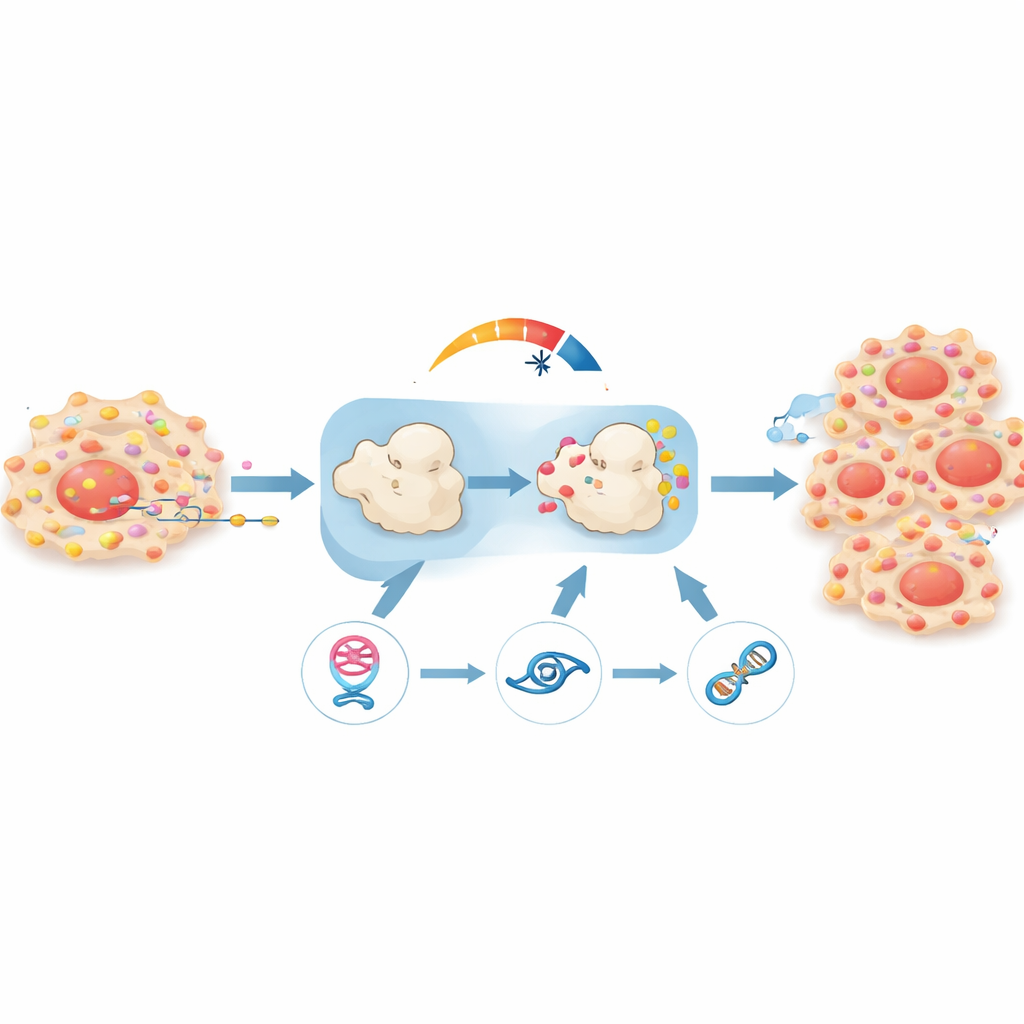
Evidence from animal models
To test whether this loop truly matters in a living organism, the team implanted human prostate cancer cells into mice. Tumors in which circPHGDH had been silenced grew more slowly, weighed less, and showed fewer signs of spread, as measured by whole-body imaging and tissue analysis. Markers of cell proliferation were lower, and the tumor structure looked less aggressive under the microscope. When the researchers either blocked miR-149 or forced RAP1B back up in these same tumors, much of the growth and spread returned, confirming that the circPHGDH–miR-149–RAP1B chain is a central driver of disease behavior.
What this means for future treatments
Together, the findings reveal a positive feedback loop in which a splicing protein, a circular RNA, a small regulatory RNA, and a signaling protein cooperate to push prostate cancer cells toward rapid growth, invasion, and sugar-hungry metabolism. For non-specialists, the key message is that cancer cells can wire their genetic and metabolic controls into self-reinforcing circuits that keep the disease progressing. Breaking this loop—by disrupting circPHGDH, restoring miR-149, blocking RAP1B, or interfering with the lactate-dependent modification of ESRP1—offers several promising avenues for future drugs aimed at slowing or halting aggressive prostate cancer.
Citation: Wang, X., Yu, L., Qian, X. et al. A ESRP1/circPHGDH/miR-149/RAP1B positive feedback loop promotes the malignant behaviors and glycolysis of prostate cancer cell. Exp Mol Med 58, 622–635 (2026). https://doi.org/10.1038/s12276-026-01646-x
Keywords: prostate cancer, circular RNA, tumor metabolism, microRNA, signal pathways