Clear Sky Science · en
PAK4 in metabolic diseases: regulation by nutrient signals and therapeutic implications
Why a Cell Switch Matters for Everyday Health
Obesity, type 2 diabetes and fatty liver disease are often blamed on calories alone, but deep inside our cells, molecular switches decide whether we burn or store fuel. This review focuses on one such switch, a protein called PAK4, and explains how it responds to changing nutrient and hormone signals, reshaping metabolism in fat, liver and muscle. Understanding this hidden control system may open the door to new treatments that tackle several metabolic diseases at once rather than one symptom at a time. 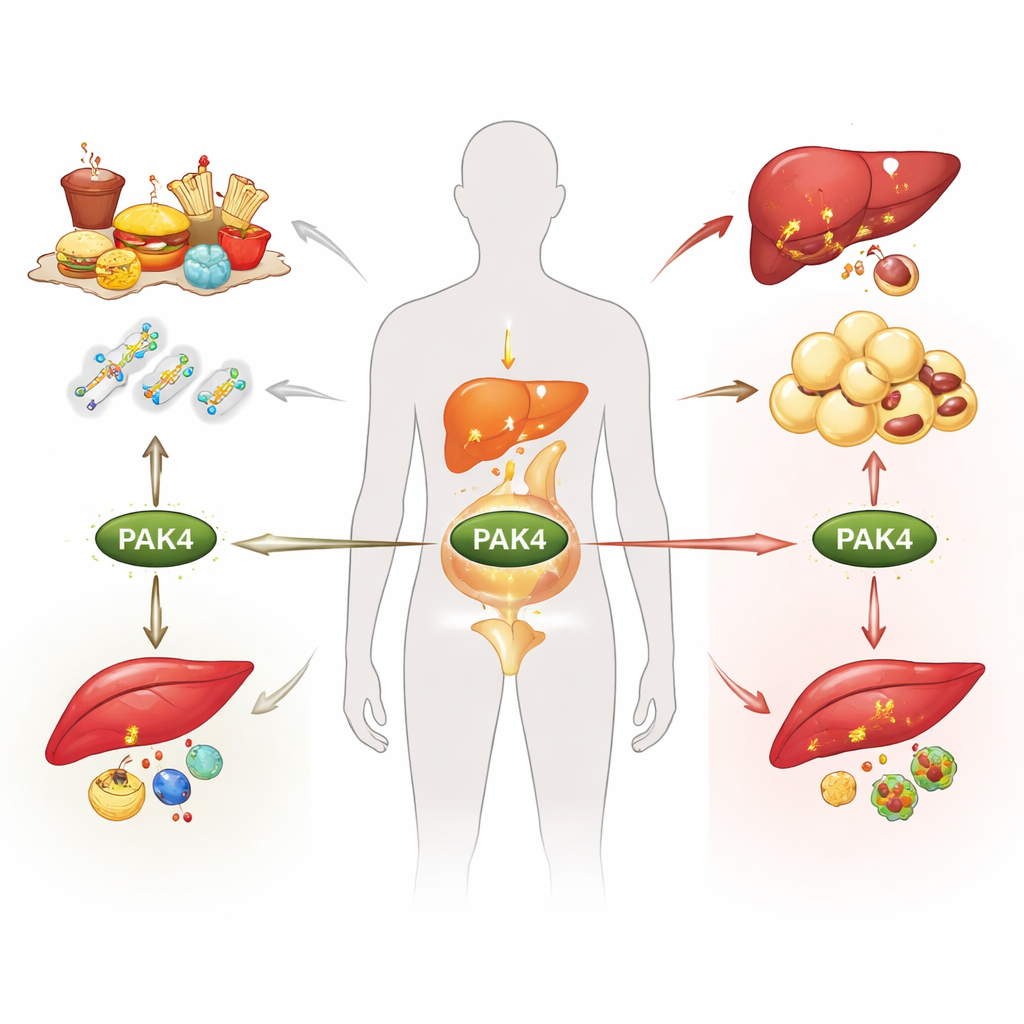
A Master Switch Hidden in Our Cells
PAK4 belongs to a family of enzymes that relay signals from small molecular “messengers” inside cells. Under healthy conditions, PAK4 is present at low levels in major metabolic organs. When energy balance is disturbed, as in obesity or type 2 diabetes, its levels rise in fat tissue, liver and skeletal muscle. PAK4 activity is controlled in several ways: by upstream messengers that change its shape, and by chemical tags added or removed after the protein is made. These include phosphate tags that can either turn PAK4 on or mark it for destruction, as well as tags that affect how stable or active it is. Fasting, feeding, hormones and stress all influence these tags, so PAK4 effectively “senses” the body’s nutritional state and passes that information on to key metabolic pathways.
How PAK4 Tilts Fat Cells Toward Storage
In fat cells, PAK4 acts as a brake on fat burning. Normally, when we fast or when stress hormones rise, another enzyme called PKA triggers the controlled breakdown of stored fat inside tiny droplets. The review describes how PAK4 counters this process by placing inhibitory phosphate tags directly on hormone-sensitive lipase and on a fatty acid binding protein that helps shuttle fat molecules. These changes weaken the machinery that releases fat from storage. PAK4 also supports the formation of new fat cells earlier in life by helping cell cycle proteins drive precursor cells to become mature fat-storing cells. When PAK4 is removed or blocked in animal models, fat cells burn more fuel, show features of “browning” that increase energy use, and animals are protected from diet-induced weight gain. 
PAK4’s Role in Fatty Liver and Muscle Sugar Handling
In the liver, PAK4 again favors storage over burning. During fasting or on a ketogenic diet, PAK4 levels fall, allowing liver cells to ramp up fat breakdown and ketone production, which supply energy to other tissues and may even slow tumor growth. When PAK4 is abundant, it adds phosphate tags to a nuclear corepressor protein that then clamps down on PPARα, a master regulator of fat oxidation and ketone formation. The result is more fat trapped in the liver and fewer protective ketones in the bloodstream. In skeletal muscle, PAK4 interferes with AMPK, a central energy sensor that promotes both sugar uptake and mitochondrial activity. By tagging AMPK in a way that blocks its activation, PAK4 lowers the amount of glucose transporter at the muscle cell surface and contributes to insulin resistance. Muscle-specific loss of PAK4 in mice reverses these effects, improving blood sugar control even under obese conditions.
Stress, Protection and Links to Other Cell Switches
The review also highlights PAK4’s impact beyond everyday metabolism. During episodes of reduced blood flow and sudden reperfusion in organs such as the liver, PAK4 weakens antioxidant defenses by tagging and destabilizing Nrf2, a key protector against oxidative stress. At the same time, another family member, PAK1, often plays more supportive roles in heart muscle, skeletal muscle and insulin-secreting cells of the pancreas, helping maintain normal glucose handling. This contrast between harmful PAK4 signaling in metabolic disease and generally beneficial PAK1 actions underscores the need for highly selective drugs that shut down PAK4 without disturbing related proteins that are essential for heart and endocrine health.
Turning a Discovery into Treatment
Because PAK4 is overactive in cancer and in metabolic disorders, drug designers have pursued small molecules that block its activity, as well as new “degrader” drugs that tag PAK4 itself for removal. Early PAK4-blocking compounds showed anti-tumor potential but ran into challenges with specificity and drug behavior in the body. More recent molecules that more precisely target PAK4 have produced striking results in obese mice: lower body weight without less eating, healthier livers, more active muscles and improved blood sugar control. Degrader drugs go one step further by physically eliminating PAK4, and early animal studies suggest they may protect against muscle loss and certain cancers. Together, these findings support a picture in which dialing down PAK4 could simultaneously ease obesity, diabetes and fatty liver disease, turning a once obscure cell switch into a promising focus for future metabolic therapies.
Citation: Bang, I.H., Park, BH. & Bae, E.J. PAK4 in metabolic diseases: regulation by nutrient signals and therapeutic implications. Exp Mol Med 58, 416–424 (2026). https://doi.org/10.1038/s12276-026-01645-y
Keywords: PAK4, metabolic disease, obesity, fatty liver, insulin resistance