Clear Sky Science · en
The redox-sensitive protein HMGB1: intracellular and extracellular roles
Why a Shape-Shifting Protein Matters for Health
Inside almost every cell in your body sits a small protein called HMGB1 that behaves a bit like a multitool. Under calm conditions it quietly helps organize DNA. But when cells are stressed or damaged, HMGB1 can move out of the nucleus, slip into the surrounding tissue, and act as an alarm signal for the immune system. This review article explains how tiny chemical changes driven by oxidation turn HMGB1 into very different biological personalities—ones that can protect tissues, drive inflammation, or even contribute to chronic disease. Understanding this molecular "mood ring" may open new ways to calm excessive inflammation without shutting the immune system down entirely.
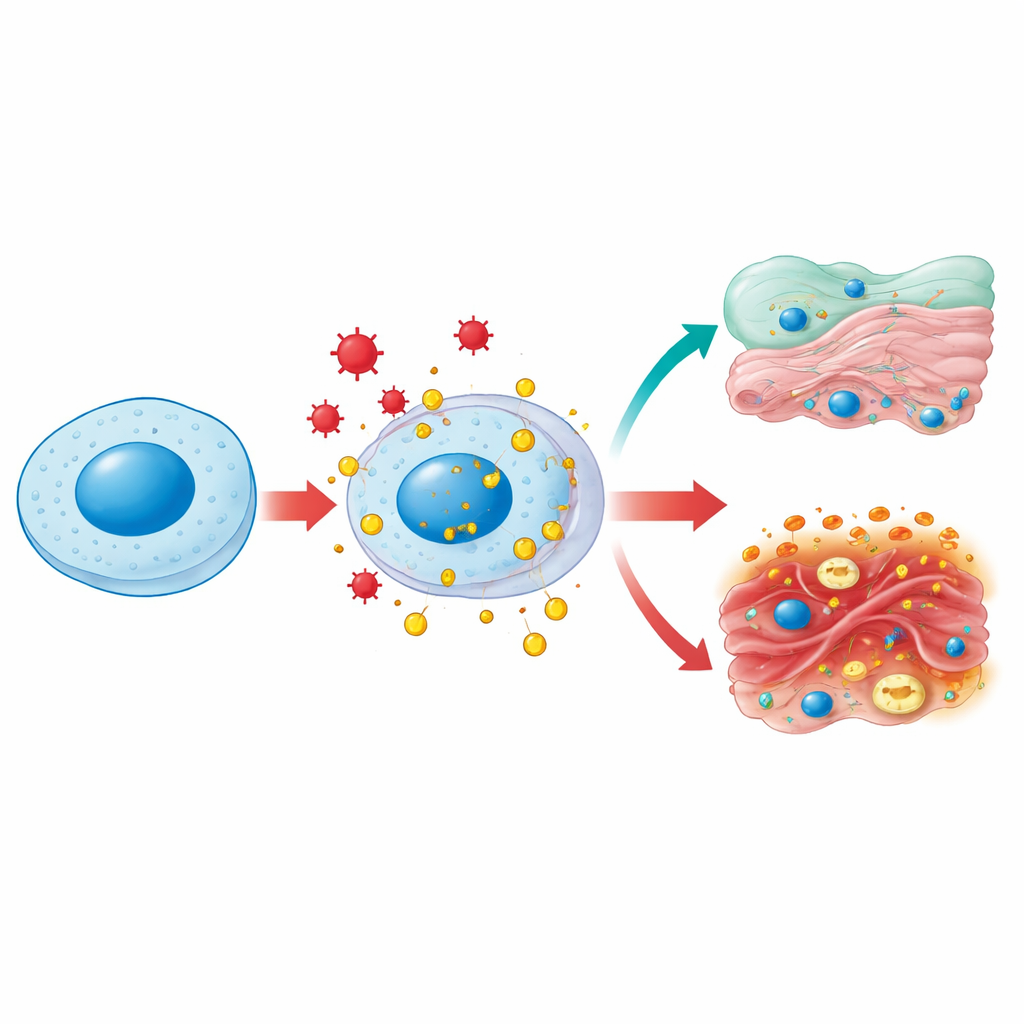
One Protein, Many Neighborhoods
HMGB1 normally lives in the cell nucleus, where it bends and loosens DNA so genes can be copied and repaired. Under stress—such as infection, high blood sugar, or toxic injury—it can be chemically modified and escorted out to the cell’s watery interior and then to the outside world. In the cytoplasm, HMGB1 helps keep mitochondria, the cell’s power plants, healthy by encouraging a clean-up process called autophagy and by preventing excessive fragmentation of these organelles. When released outside the cell, HMGB1 becomes a classic “danger signal”: immune cells recognize it as a sign that tissues have been injured and rush to the scene. The same molecule therefore plays housekeeping roles inside cells and alarm-sounding roles outside them, depending on where it is and how it has been chemically tuned.
How Oxidation Rewrites Its Behavior
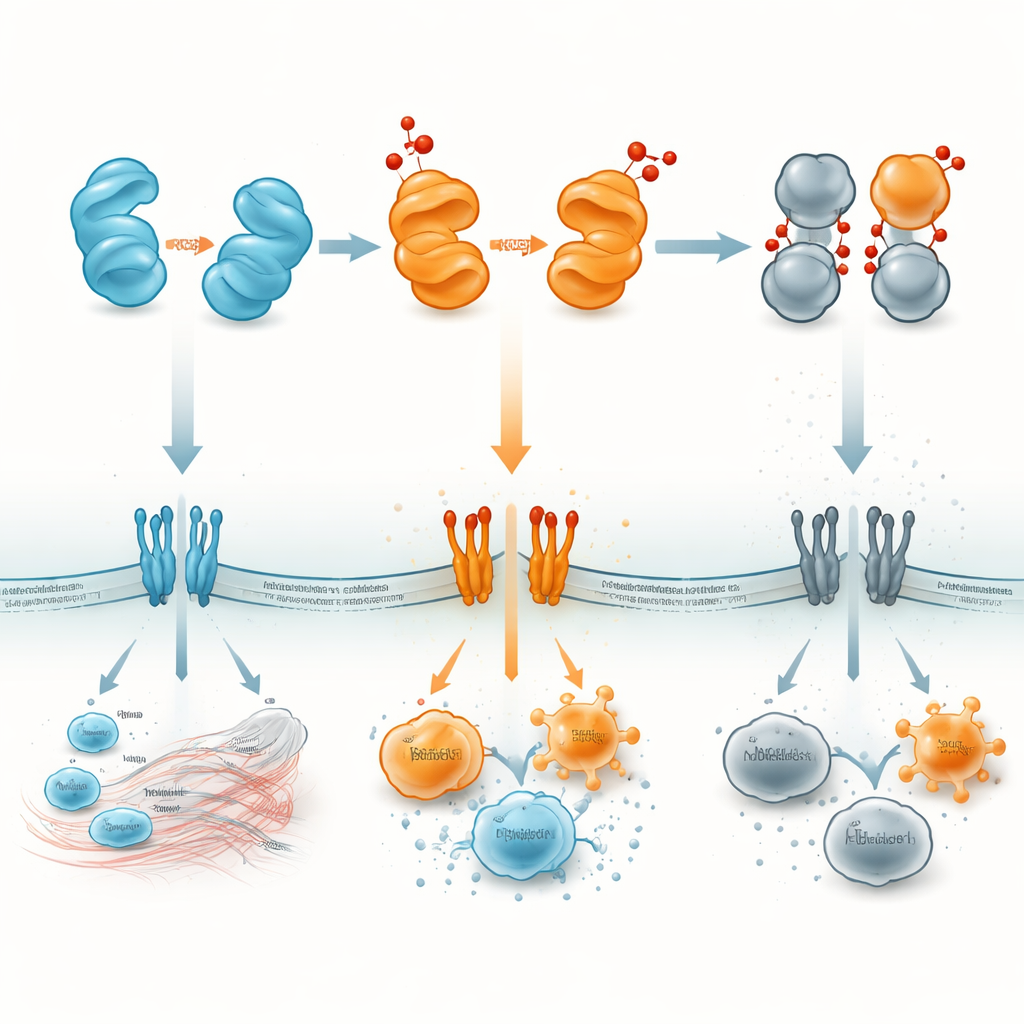
The centerpiece of this review is how oxidation—reactions driven by reactive oxygen species (ROS)—reshapes HMGB1. The protein contains three sulfur-bearing building blocks that act as tiny switches. In their fully reduced state, HMGB1 tends to support cell survival and the recruitment of repair cells. Mild oxidation can link two of these sites together within the same protein, creating a “disulfide” form that is especially good at latching onto immune receptors such as Toll-like receptors and RAGE on cell surfaces. This version strongly stimulates the production of inflammatory messengers. Stronger oxidative stress can push the molecule further into an over-oxidized form that can no longer bind DNA or receptors effectively; this “burned-out” version becomes immunologically silent and is associated with the resolution phase of inflammation and cell death programs that do not provoke an immune attack.
From Cell Death Signals to Disease Drivers
HMGB1 is deeply entwined with the many ways cells die. During violent forms of death such as necrosis, necroptosis, ferroptosis, and pyroptosis, HMGB1 leaks or is actively transported out of cells, carrying a redox signature that reflects the surrounding oxidative conditions. Early in necrosis, it tends to be in a reduced form, while prolonged stress pushes it toward more oxidized states. Once outside, reduced HMGB1 can foster protective autophagy in some cancer cells, helping them survive chemotherapy, whereas disulfide-rich and dimerized forms amplify inflammatory cascades and complement activation that can worsen tissue damage. HMGB1 can also feed back to promote certain death pathways—for example, by partnering with bacterial lipids to trigger necroptosis or by driving iron-dependent cell death in brain support cells after stroke. In this way, HMGB1 both marks and modulates the life–death balance in tissues.
Links to Inflammation, Autoimmunity, and Cancer
Because its behavior is so sensitive to oxidation, different HMGB1 variants show up in distinct patterns across diseases. Disulfide-rich HMGB1 is strongly associated with chronic inflammation: it appears in inflamed joints in rheumatoid arthritis, in fibrotic liver, in lung injury, in ischemia–reperfusion damage after surgery, and in sepsis, where its blood levels correlate with severity and death risk. Reduced HMGB1, by contrast, often tracks with cell migration and tissue remodeling, such as the movement of heart fibroblasts or monocytes guided by the chemokine CXCL12. Over-oxidized, immunologically quiet HMGB1 is enriched in settings like late-stage apoptosis, where the body wants to clear dying cells without provoking an attack. In tumors, the disulfide form supports a suppressive, growth-permissive immune environment, while blocking HMGB1 can shrink tumors and make checkpoint immunotherapies more effective.
Turning a Molecular Alarm into a Therapeutic Target
To a lay observer, HMGB1 can be thought of as a small protein alarm whose volume and message change with its chemical state. Reduced HMGB1 tends to call in repair crews; the disulfide and dimer forms can blare a harsh alarm that fuels damaging inflammation; and the fully oxidized form effectively falls silent, helping inflammation wind down. By mapping where and when each version appears—inside nuclei, in the cytoplasm, in blood, or in specific organs—researchers hope to design drugs that either block the harmful forms or stabilize the helpful ones. Such strategies might allow doctors to treat sepsis, autoimmunity, stroke, lung injury, or cancer by targeting not just HMGB1 itself, but the oxidative “dimmer switch” that controls how this protein speaks to the immune system.
Citation: Kwak, M.S., Jung, S.F., Park, I.H. et al. The redox-sensitive protein HMGB1: intracellular and extracellular roles. Exp Mol Med 58, 345–356 (2026). https://doi.org/10.1038/s12276-026-01640-3
Keywords: HMGB1, inflammation, oxidative stress, cell death, autoimmune disease