Clear Sky Science · en
Mechanotransduction through T cell receptors: consensus, controversies and future outlooks
How Immune Cells Feel Their World
Our immune system does more than sniff out chemical signals; it also feels physical forces. This review article explores how T cells — white blood cells that hunt viruses and cancer — may use tiny pushes and pulls on their surface receptors to decide whether to attack. Understanding this "sense of touch" at the molecular scale could reshape how we think about vaccines, cancer immunotherapy and autoimmune disease.
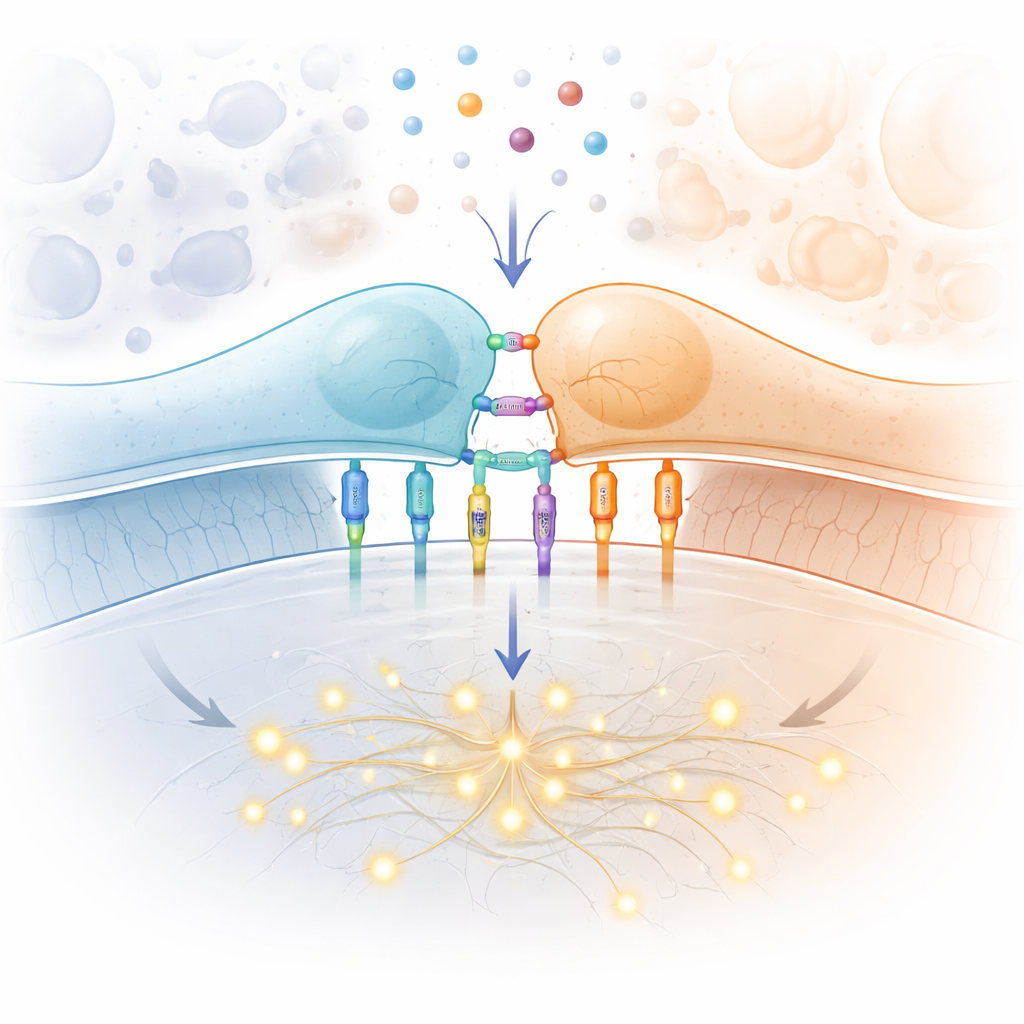
The Special Job of the T Cell Receptor
At the heart of the story is the T cell receptor, a molecular machine on the T cell surface that inspects fragments of proteins displayed by other cells. When the right fragment is found, the T cell can spring into action, multiply and kill infected or cancerous targets. This receptor must meet a tough set of demands: it has to react to vanishingly few foreign fragments, ignore a sea of harmless self fragments, work quickly as T cells patrol the body and do so for millions of different receptor variants in each person. The classic wiring of the signaling pathway inside the cell — involving protein kinases, scaffolds and transcription factors — is well mapped out. What remains mysterious is the very first step: how the simple act of binding to a fragment at the cell surface flips the receptor from silent to active.
Competing Ideas for the First Spark
Researchers have proposed several models for how the T cell receptor first turns on. In one view, signaling begins when receptors are brought together into small clusters, boosting local concentration and allowing signaling reactions to proceed more efficiently. Another idea emphasizes shape changes: binding could twist or relax parts of the receptor complex, freeing buried segments inside the cell so that enzymes can modify them. A third model highlights crowding at the cell–cell contact. Here, short receptor–ligand pairs pack into tight zones that physically exclude bulky enzymes that normally shut signals off, tipping the balance toward activation. Each model is supported by experiments and explains part of the puzzle, but none alone fully accounts for the receptor’s extreme sensitivity and its ability to distinguish between very similar protein fragments.
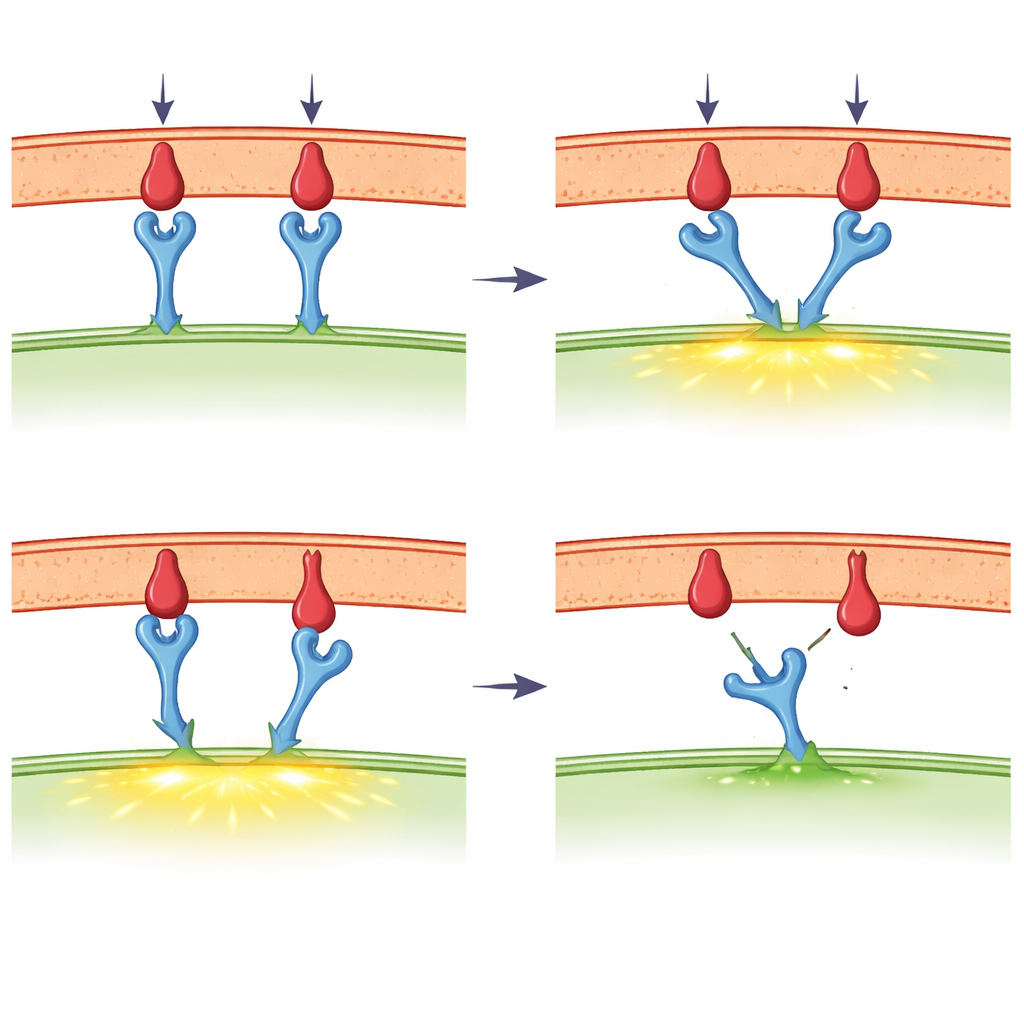
When Touch and Force Enter the Picture
A major focus of the review is the emerging idea that the T cell receptor behaves as a mechanosensor — a device that responds to force. Using ultra-sensitive instruments, scientists have pulled on single receptor–ligand bonds with forces a trillion times smaller than the weight of an apple. They find that for potent foreign fragments, a modest tug can actually prolong the lifetime of the bond, a behavior known as a "catch" response. Weaker or self fragments instead show "slip" behavior: they let go more quickly when pulled. T cells themselves generate such forces through their internal skeleton of actin filaments and motor proteins, especially at the tight contact zone called the immune synapse. New molecular tension probes show that forces in the range where catch behavior appears are indeed present during early T cell activation, though different experimental methods sometimes report different values and have sparked lively debate.
Decoding Self from Nonself Over Time
The article also revisits how T cells might turn noisy, fleeting contact events into reliable decisions. A long-standing idea, kinetic proofreading, suggests that signaling proceeds through a sequence of steps that requires time; only fragments that keep the receptor engaged long enough allow the chain to reach a point of no return. The authors discuss how mechanical effects can sharpen this time filter: pulling forces extend the lifetimes of productive bonds and shorten unproductive ones, widening the gap between strong and weak stimuli. They also consider how T cells may integrate many short contacts rather than relying on a single long one, and how feedback within the signaling network can store a kind of molecular "memory" of recent encounters. These refinements help explain how T cells achieve both speed and accuracy in complex tissue environments.
Shared Principles Across Immune Receptors
Although the T cell receptor is unusually demanding in what it must accomplish, many of its design principles appear in other immune receptors. B cell receptors and antibody receptors on innate immune cells share similar signaling motifs and often operate in close-contact zones where crowding, clustering and cytoskeletal forces all matter. Catch-like responses to force have now been reported for several such receptor–ligand pairs. This suggests that feeling mechanical cues may be a general strategy the immune system uses to check whether a target is firmly anchored, correctly presented and worth responding to.
Why This Matters for Health and Therapy
For a lay reader, the takeaway is that T cells do not simply smell the presence of foreign molecules — they also test how those molecules feel when pulled. By combining chemistry, physics and cell biology, this review argues that immune receptors convert subtle differences in both binding and force into life-or-death decisions for cells. A deeper grasp of these mechanobiological rules could guide the design of better T cell–based therapies, more precise vaccines and new treatments that tune immune responses up or down by altering not just what receptors bind, but how they are mechanically engaged.
Citation: Travaglino, S., Jeon, Y., Kim, Y. et al. Mechanotransduction through T cell receptors: consensus, controversies and future outlooks. Exp Mol Med 58, 319–335 (2026). https://doi.org/10.1038/s12276-026-01639-w
Keywords: T cell receptor, mechanotransduction, catch bonds, immune synapse, kinetic proofreading