Clear Sky Science · en
UBE2M as a bridge spanning neddylation and cell cycle regulation in colorectal adenocarcinoma
Why this research matters for bowel cancer
Colorectal cancer is one of the most common and deadly cancers worldwide, largely because tumor cells learn to divide relentlessly. This study uncovers a previously hidden control switch that helps colorectal cancer cells race through a critical checkpoint of cell division. By revealing how this switch works at the molecular level—and showing that an existing antifungal drug can interfere with it—the work points to a potential new way to slow or stop tumor growth.
A hidden layer of control inside tumor cells
Cells do not divide at random; they move through an orderly cycle with checkpoints that confirm whether it is safe to copy DNA and split in two. Cancer cells often tamper with these checkpoints. The authors focused on a chemical tagging process called neddylation, which subtly alters proteins after they are made. Earlier studies hinted that neddylation is more active in many cancers, but how it connects to the division cycle of colorectal cancer cells was unclear. Using large single-cell datasets from human tumors, along with bulk gene-expression data from more than 1,800 patients, the team found that neddylation activity is especially high in malignant bowel cells that are poised in the G2/M phase—the final gate before a cell divides.
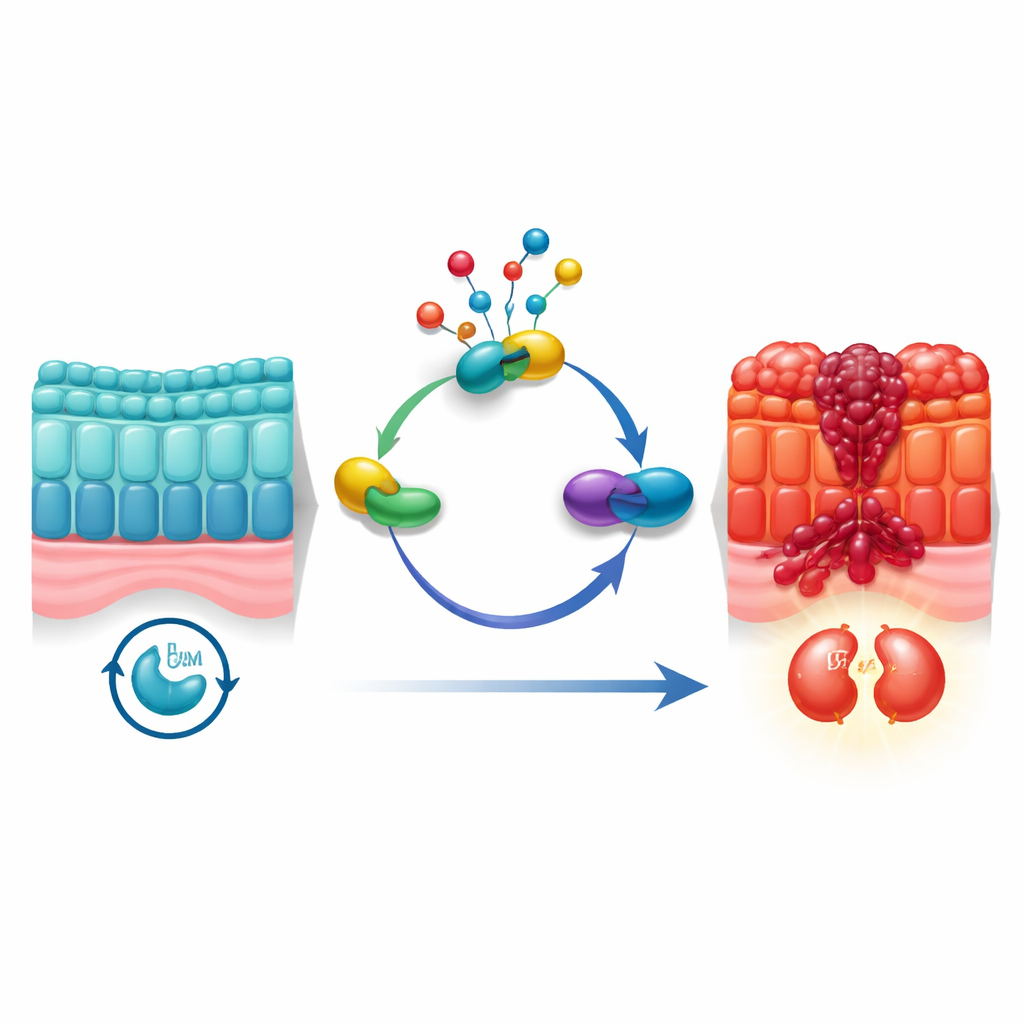
Spotlight on a molecular “bridge” protein
To work out which molecules connect neddylation to cell division, the researchers used network-style computational analyses to sift through thousands of genes. One protein stood out: UBE2M, an enzyme that helps attach the small NEDD8 tag to other proteins. UBE2M was not only strongly linked to neddylation and cell-cycle pathways in the data; it was also found at much higher levels in colorectal tumors than in nearby normal tissue. Patients whose tumors had more UBE2M tended to have worse survival, marking it as a potential driver rather than a bystander. When the team reduced UBE2M in cancer cell lines and in mouse tumor models, tumor growth slowed, cells divided less, and more cells underwent programmed death. Conversely, forcing cells to make extra UBE2M sped up growth and cell-cycle progression.
A relay that protects a key growth-promoting protein
Diving deeper, the scientists asked how UBE2M helps cells divide. They discovered that UBE2M does not act alone but sets off a relay involving two other proteins, USP39 and PABPC1. Normally, PABPC1 is marked for destruction by a chain of ubiquitin tags, which send it to the cell’s protein recycling machinery. The team showed that UBE2M chemically modifies USP39 with the NEDD8 tag. This modification boosts USP39’s ability to snip ubiquitin chains off PABPC1, rescuing it from destruction and making it more stable. With more PABPC1 around, the cell becomes better at translating specific messenger RNAs into protein, including the mRNA for CCNB1, a cyclin that acts as a gas pedal for the G2/M checkpoint. In essence, UBE2M helps stabilize PABPC1, which in turn boosts production of CCNB1, pushing cells more readily into division.
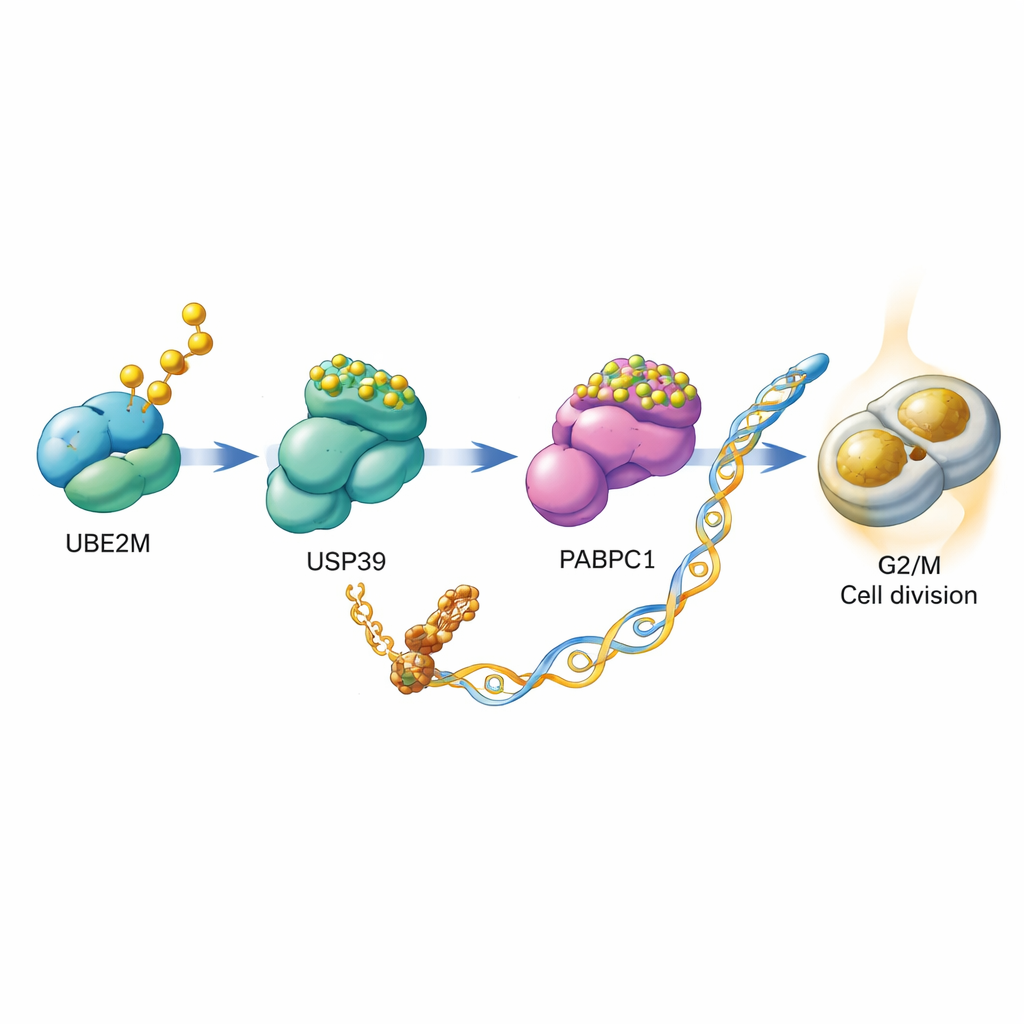
From molecular mechanism to drug opportunity
The discovery of this UBE2M–USP39–PABPC1–CCNB1 chain suggested a new vulnerability in colorectal cancer cells. The authors turned to micafungin, a drug already used to treat fungal infections and recently identified as an inhibitor of UBE2M’s neddylation activity. In bowel cancer cells grown in the lab, micafungin weakened USP39 neddylation, increased the degradation of PABPC1, and lowered CCNB1 protein levels. As a result, cell division slowed, more cells stalled at the G2/M checkpoint, and apoptosis increased. In mice implanted with colorectal cancer cells, daily micafungin treatment significantly shrank tumors compared with untreated animals, without needing to remove UBE2M itself.
What this means for patients in the long run
This work paints a clear picture of how a single enzyme, UBE2M, can link a subtle protein-tagging system to the decision of a bowel cancer cell to divide. By stabilizing a translation factor (PABPC1) via USP39, UBE2M indirectly raises levels of a powerful cell-cycle driver, CCNB1, allowing tumors to grow faster. Although more research and clinical testing are needed, the findings suggest that blocking this relay—potentially with repurposed drugs like micafungin—could offer a new targeted strategy to slow colorectal cancer progression and improve outcomes for patients.
Citation: Wang, Z., Wang, Y., Chen, Y. et al. UBE2M as a bridge spanning neddylation and cell cycle regulation in colorectal adenocarcinoma. Exp Mol Med 58, 501–518 (2026). https://doi.org/10.1038/s12276-026-01636-z
Keywords: colorectal cancer, cell cycle, neddylation, UBE2M, targeted therapy