Clear Sky Science · en
Drug-induced gastrointestinal toxicity and barrier integrity: cytoskeleton-mediated impairment in a clinically relevant human intestinal epithelium model
Why Gut Side Effects Matter
Many medicines that fight cancer, inflammation or other illnesses can unintentionally harm the lining of our intestines. When this inner “skin” of the gut is damaged, people can suffer from diarrhea, pain, nausea and poor nutrient absorption, sometimes so severe that doctors must stop or lower life‑saving treatments. Yet today’s lab tests often miss these problems before drugs reach patients. This study introduces a more realistic laboratory model of the human intestine and shows how it can spot gut‑damaging drugs earlier and reveal how they weaken the body’s natural barrier.
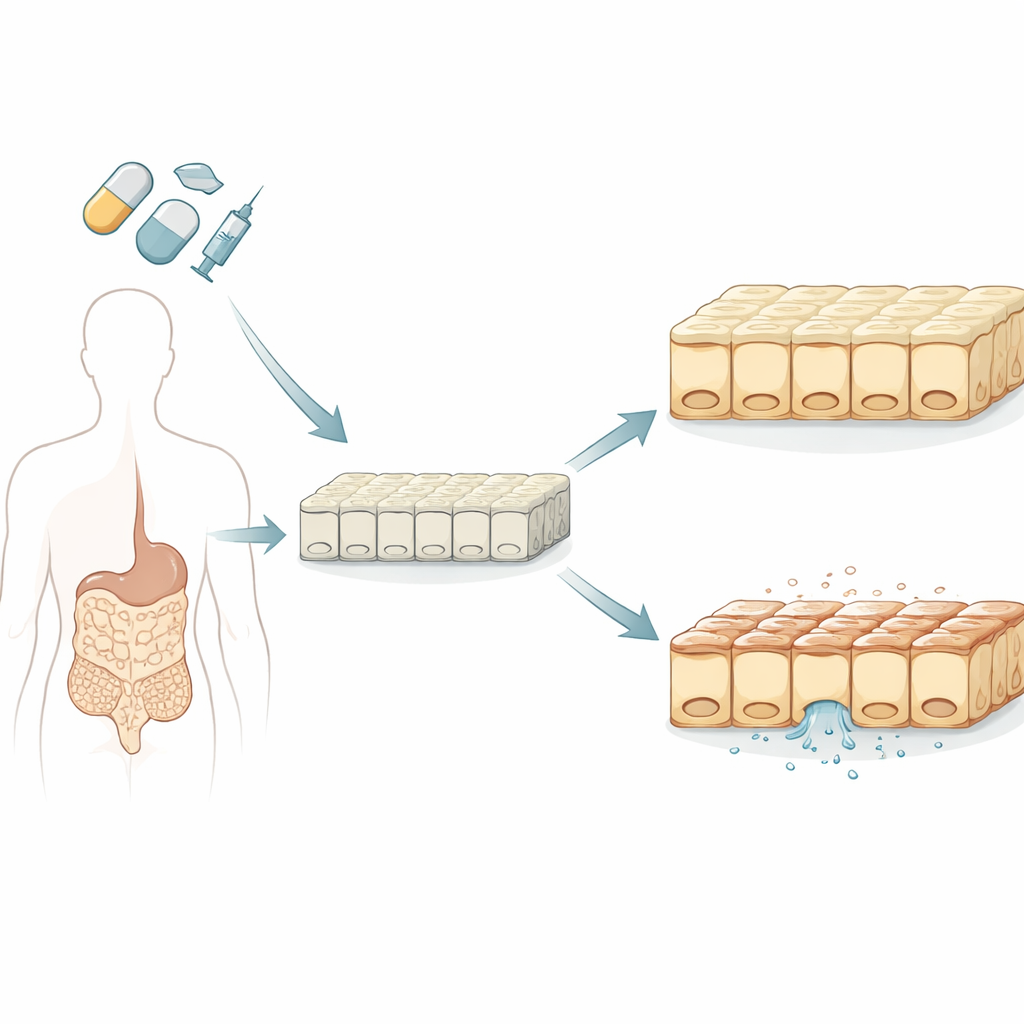
Building a Better Mini-Gut
The researchers began by growing sheets of human intestinal cells in the lab using stem cells, which can develop into many tissue types. Unlike a traditional cancer‑derived cell line long used for drug testing, these stem‑cell‑derived cells formed a mixed community that more closely resembles the real small intestine, including mucus‑secreting and hormone‑producing cells. The team confirmed that these lab‑grown linings had realistic leak‑tightness, proper top‑and‑bottom polarity and key transport and metabolism features that influence how medicines move through and are processed by the gut.
Measuring the Electrical Tightness of the Barrier
To test gut safety, the group focused on transepithelial electrical resistance, or TEER, a noninvasive readout of how well neighboring cells seal together. High TEER means a tight, protective barrier; falling TEER means the wall between the gut and the bloodstream is becoming leaky. The scientists compared TEER with a standard cell‑survival test that measures energy molecules such as ATP. They exposed both their new intestinal model and the older cancer cell model to 17 drugs known to differ in how often they cause gastrointestinal side effects, including common chemotherapy agents, targeted cancer pills and painkillers like ibuprofen and other anti‑inflammatory drugs.
Finding Hidden Damage Before Cells Die
Across this panel of medicines, TEER measurements in the stem‑cell‑derived gut lining outperformed the traditional ATP test and the older cancer cell barrier. Several chemotherapy drugs showed little change in ATP, suggesting cells were still alive, yet they caused large drops in TEER and obvious damage in live‑dead staining images. This means the barrier can fail before cells fully die, an early warning that classic viability tests miss. When the team compared their lab results with clinical records of how often each drug triggers gut symptoms, the new TEER assay correctly flagged almost all of the higher‑risk drugs and correctly reassured for the low‑risk ones, achieving a very high accuracy.
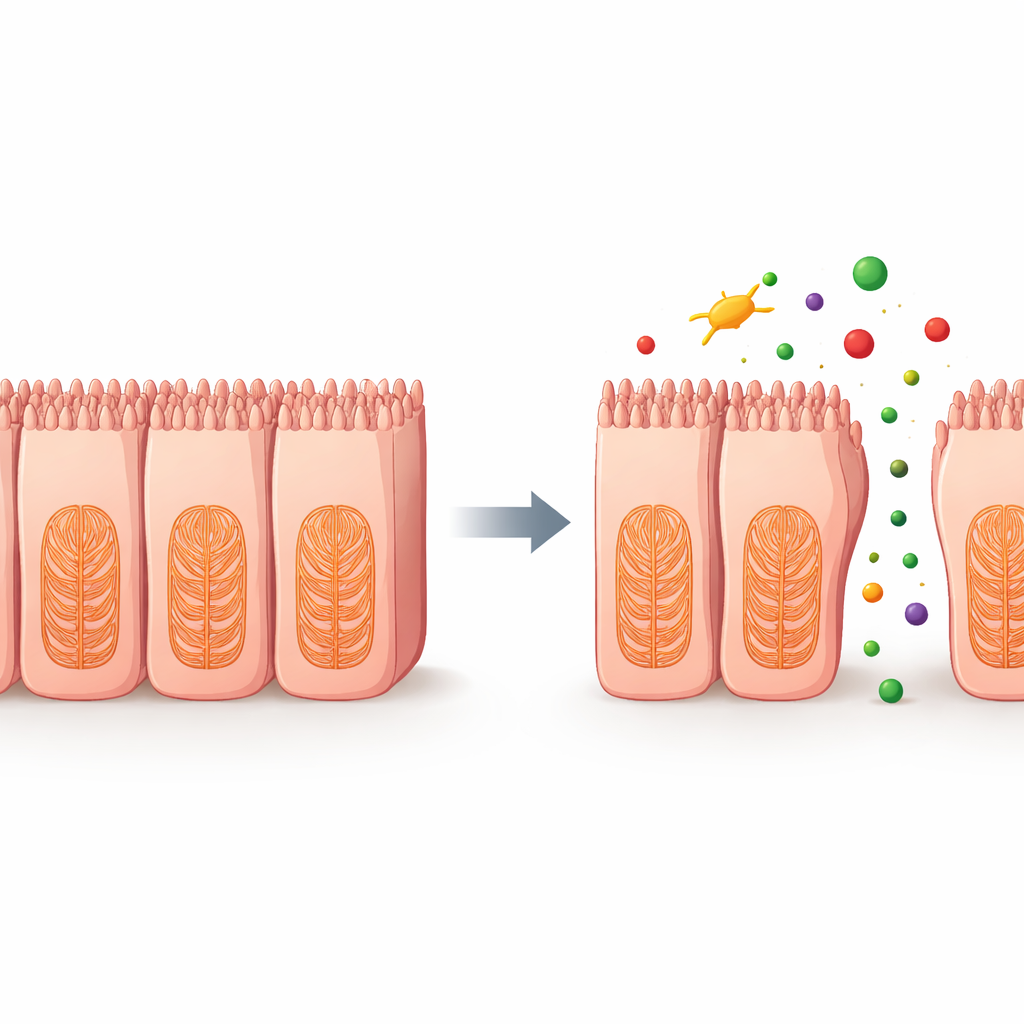
How Drugs Undermine the Inner Scaffold
To uncover what was going wrong inside the cells, the scientists analyzed gene activity after treatment with two chemotherapy agents that target microtubules, a key part of the cell’s internal scaffold. They found broad reductions in genes involved in the cytoskeleton, cell‑to‑cell adhesion and the extracellular matrix—the meshwork that helps cells anchor to their surroundings. Additional experiments showed dose‑dependent increases in reactive oxygen species, unstable oxygen‑containing molecules that can damage cellular structures. Together, these changes point to a chain of events in which certain drugs disturb the inner framework and connections of intestinal cells, loosening the barrier and allowing substances to leak between cells.
What This Means for Patients
This work shows that a stem‑cell‑based human intestinal lining, paired with simple electrical measurements of barrier tightness, can more reliably predict which medicines are likely to cause gut injury than long‑standing lab methods. By detecting barrier weakening early—and tying it to underlying damage to the cellular scaffold—this platform could help drug developers discard or redesign risky compounds before they ever reach the clinic. In the long run, such realistic “mini‑gut” tests may reduce unpleasant or dangerous gastrointestinal side effects for patients while allowing doctors to use powerful therapies more safely.
Citation: Yu, W.D., Lee, S., Cho, HS. et al. Drug-induced gastrointestinal toxicity and barrier integrity: cytoskeleton-mediated impairment in a clinically relevant human intestinal epithelium model. Exp Mol Med 58, 487–500 (2026). https://doi.org/10.1038/s12276-025-01635-6
Keywords: gastrointestinal toxicity, intestinal barrier, stem cell model, drug safety, chemotherapy side effects