Clear Sky Science · en
CDK13 drives clear cell renal carcinoma through METTL16-mediated m6A modification of ACLY mRNA
Why Fat-Filled Kidney Tumors Matter
Clear cell kidney cancer often looks pale and oily under the microscope because its cells are crammed with fat. That unusual appearance is not just cosmetic; it reflects a deeper rewiring of how these cancer cells handle fuel. This study asks a simple but important question: what molecular switch tells kidney tumor cells to hoard fat, and can that switch be turned off to slow the disease?
A Hidden Conductor of Tumor Growth
The researchers focused on a protein called CDK13, part of a family of enzymes that normally help cells divide. By analyzing large patient datasets and tumor samples, they found that CDK13 levels are consistently higher in clear cell kidney cancers than in normal kidney tissue. Patients whose tumors carried more CDK13 tended to have larger, more advanced cancers and poorer outcomes. When the team lowered CDK13 in kidney cancer cell lines, the cells grew more slowly and had trouble moving through the cell cycle, suggesting that CDK13 acts as a hidden conductor coordinating both growth and survival.
From Sugar to Fat: Rewiring the Cell’s Fuel Factory
Because clear cell kidney tumors are packed with lipids, the team examined whether CDK13 also controls how these cells make fat. Using a combination of gene-expression profiling and microscopic fat stains, they showed that ramping up CDK13 increases the build-up of lipid droplets inside cancer cells, while reducing CDK13 has the opposite effect. CDK13 strongly influenced an enzyme called ACLY, which converts a common metabolic intermediate into acetyl-CoA, the starting material for building fatty acids and cholesterol. High CDK13 went hand in hand with high ACLY in patient tumors, and both proteins were concentrated in the same regions of cancer tissue. When ACLY was artificially boosted, it rescued many of the growth and fat-storage defects caused by CDK13 loss, placing ACLY as a key downstream effector of this pathway. 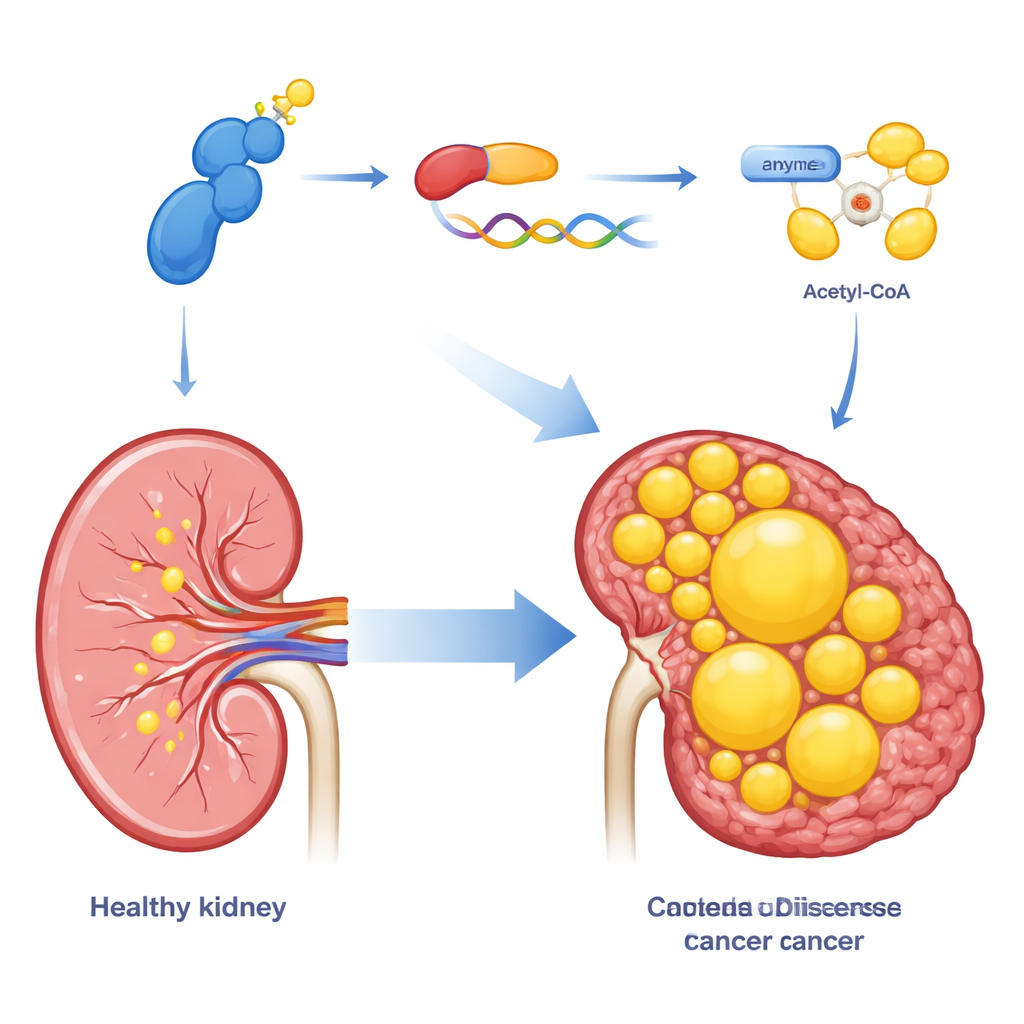
A Layered Message System Inside Cancer Cells
Rather than acting directly on ACLY as a simple on–off switch, CDK13 exerts its power through a layered message system built on RNA, the molecule that carries genetic instructions from DNA to protein factories. The authors discovered that CDK13 physically binds to and chemically modifies another enzyme, METTL16, which decorates specific RNA messages with tiny chemical marks called methyl groups. CDK13 adds a phosphate tag to METTL16 at a precise spot, making METTL16 more active. In turn, METTL16 places extra methyl marks on the RNA blueprint for ACLY. These marks do not change the genetic code itself, but they alter how the cell treats that message. A third protein, YTHDC2, recognizes the marked ACLY RNA and protects it from decay, allowing more ACLY protein to be produced over time. This cascade—CDK13 activating METTL16, METTL16 marking ACLY RNA, and YTHDC2 guarding that marked message—creates a powerful feed to drive fat synthesis. 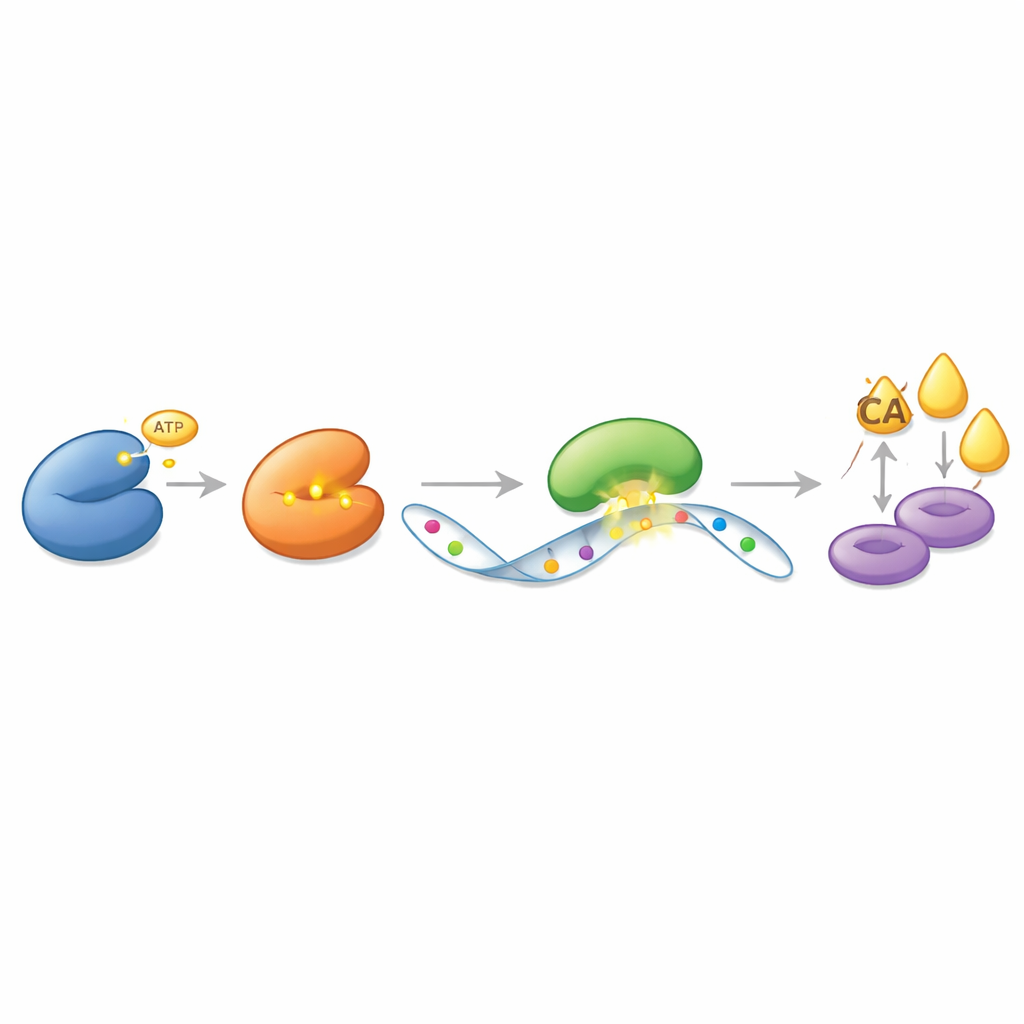
Testing the Chain in Cells, Mice, and Patient Samples
The strength of the work lies in how thoroughly the authors tested this chain of events. In cultured kidney cancer cells, disrupting any part of the CDK13–METTL16–ACLY pathway reduced lipid droplets and slowed proliferation. In mice implanted with human kidney cancer cells, blocking CDK13 or ACLY alone shrank tumors and cut down fat stores within them, while blocking both together had an even stronger effect. The team also used a small-molecule compound, 1NM-PP1, which inhibits CDK13 activity. This drug-like agent reduced the activating tag on METTL16, lowered ACLY levels, and suppressed tumor growth, especially when combined with METTL16 depletion. Across patient datasets, CDK13, METTL16, and ACLY tended to rise and fall together, reinforcing the idea that this axis is active in real cancers, not just in lab models.
What This Could Mean for Future Treatments
For non-specialists, the key message is that this study exposes a new control knob for the “fat factory” inside clear cell kidney tumors. Instead of targeting only the enzymes that make lipids, the researchers reveal a higher-level command chain that stabilizes the instructions for those enzymes. By interrupting the CDK13–METTL16–ACLY axis, it may be possible to starve tumors of the fats they need to grow and spread, while leaving normal cells less affected. Although the work is still preclinical and 1NM-PP1 is not yet a kidney cancer drug, the findings point toward new strategies that pair kinase inhibitors with drugs targeting RNA-modifying enzymes, offering a more precise way to treat this metabolically driven form of kidney cancer.
Citation: Chen, J., Liu, H., Zhang, Y. et al. CDK13 drives clear cell renal carcinoma through METTL16-mediated m6A modification of ACLY mRNA. Exp Mol Med 58, 472–486 (2026). https://doi.org/10.1038/s12276-025-01634-7
Keywords: clear cell renal cell carcinoma, lipid metabolism, CDK13, RNA methylation, ACLY