Clear Sky Science · en
Two codes of RNA editing by deamination in human diseases
How Cells Rewrite Their Own Messages
Every cell in your body constantly reads instructions from DNA to build and maintain you. For decades, we assumed those instructions were copied into RNA and then faithfully translated into proteins. This review article shows that the story is far less rigid: cells actually "rewrite" many RNA messages after they are made, swapping single chemical letters in ways that can subtly or dramatically change how our bodies work. Understanding this hidden layer of editing helps explain why people develop autoimmune diseases, neurological disorders, metabolic problems, infections and cancer—and how we might eventually treat them.
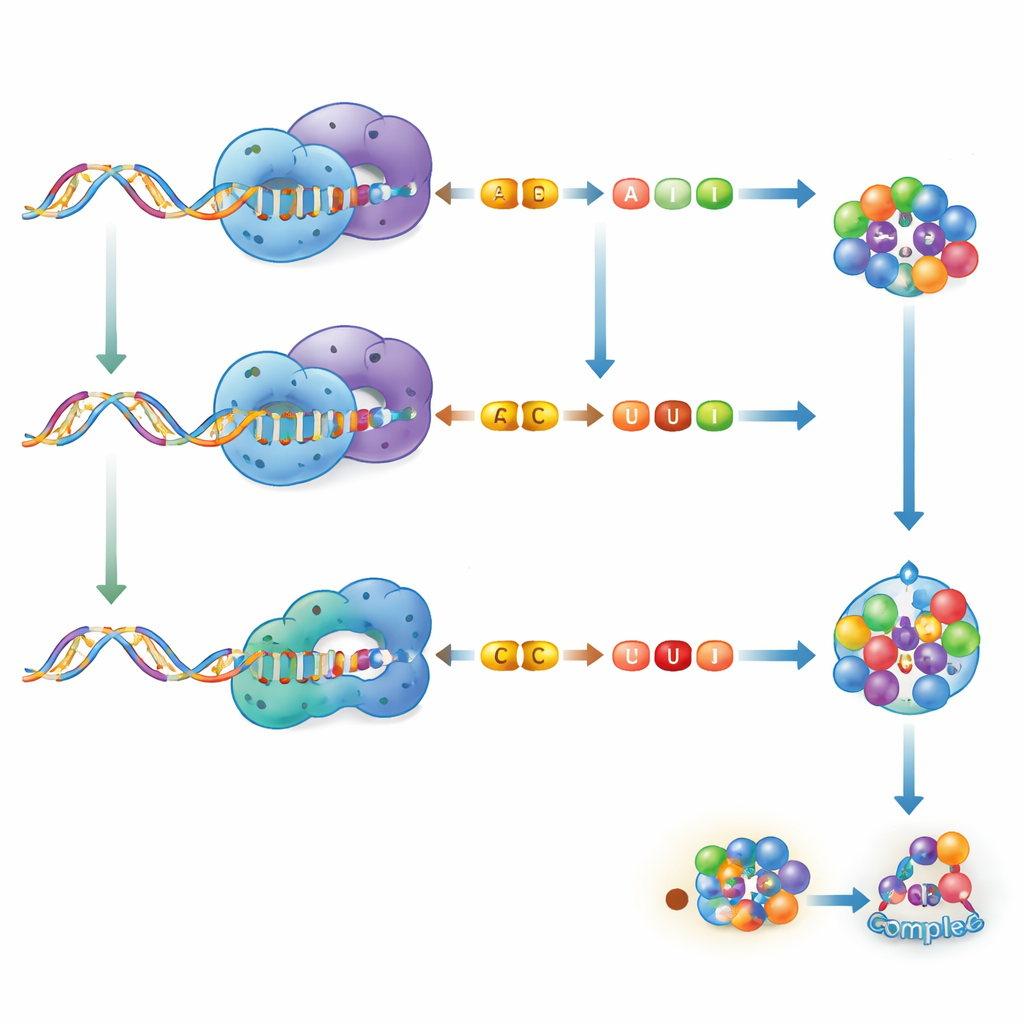
Two Ways to Change a Single Letter
The authors focus on two main kinds of RNA editing found in humans. One is called A-to-I editing, carried out by enzymes known as ADARs. They change the base adenosine (A) into inosine (I), which cellular machinery reads mostly as if it were guanosine (G). The other is C-to-U editing, handled by enzymes of the APOBEC family, which convert cytidine (C) into uridine (U). Both processes remove a small chemical group from a single base, but they differ in where they act, which RNAs they prefer, and how strongly they reshape RNA structure. A-to-I editing often alters how RNA strands pair up and can diversify proteins or change how other molecules bind RNA. C-to-U editing tends to be more subtle structurally, but still can introduce stop signals, tweak protein sequences, or fine-tune regulatory regions of RNA.
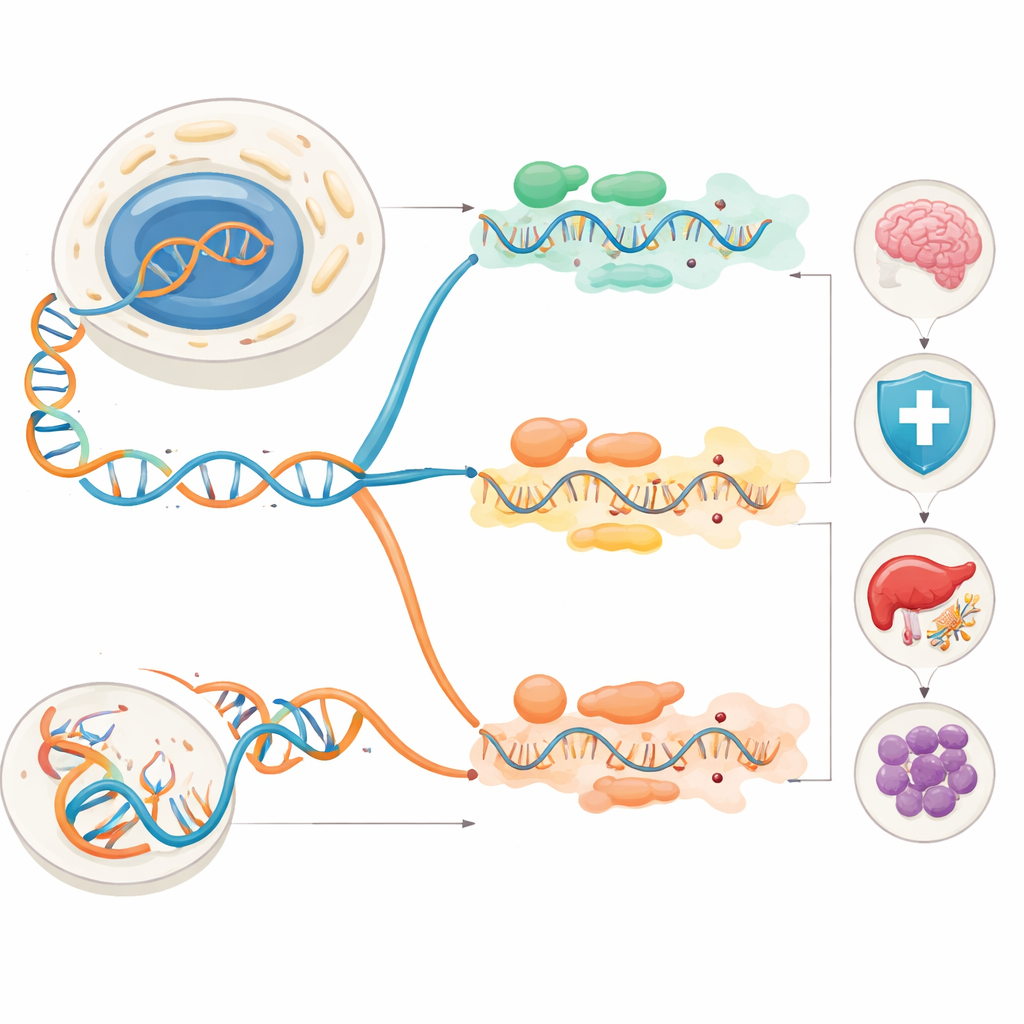
From Edited Messages to Health and Disease
Because these editing events can change protein parts or the regulatory regions around them, they touch many aspects of normal biology. In the immune system, ADAR1 edits self-made double-stranded RNAs so that viral sensors do not mistake them for foreign invaders. When ADAR1 fails, immune alarms stay stuck “on,” leading to chronic interferon signaling and autoimmune conditions such as Aicardi–Goutières syndrome. APOBEC enzymes also edit RNA in immune cells, shaping how macrophages respond to stress and inflammation and potentially contributing to diseases like systemic lupus. In the nervous system, ADAR2 editing is crucial for tuning brain receptors that control calcium flow; without it, mice suffer seizures and die early. APOBEC-driven editing of certain receptor RNAs in neurons can heighten their sensitivity and is linked to epilepsy, degeneration and cognitive problems.
Viruses, Metabolism and the Cancer Connection
RNA editing also affects how we interact with viruses, manage energy and develop cancer. ADAR1 can edit viral genomes directly, sometimes crippling the virus and sometimes, as in hepatitis delta virus, helping it complete its life cycle. APOBEC enzymes, famous for attacking retroviruses, leave a strong C-to-U signature in the RNA of SARS-CoV-2, both limiting the virus and at the same time generating mutations that may aid viral evolution. In metabolism, ADAR2 helps pancreatic beta cells adjust insulin secretion in response to diet, while ADAR1 and ADAR2 activity influences diabetes risk and fatty liver disease. APOBEC1’s classic role is to edit the RNA for apolipoprotein B, producing a shortened protein essential for transporting dietary fats; when this editing is missing, mice develop serious lipid and cholesterol problems.
How Edited RNA Shapes Tumors
The same enzymes that protect us can also drive cancer when misregulated. Large cancer sequencing projects have uncovered tens of thousands of A-to-I editing sites and widespread APOBEC-related mutations. In breast cancer, ADAR1 editing can either spur or restrain tumor behavior depending on the target RNA, affecting cell invasion, metastasis and survival. In glioblastoma, a lethal brain tumor, ADAR1 supports cancer stem cells, while ADAR2 generally acts as a brake on growth by editing both protein-coding RNAs and cancer-promoting microRNAs. In leukemias, ADAR1 often boosts malignant stem-like cells and dampens tumor-suppressive microRNAs, whereas ADAR2 edits specific targets in ways that slow disease. APOBEC-mediated C-to-U editing of certain RNAs in blood cancers can either worsen or improve patient outcomes, underscoring how context-dependent these changes are.
Unanswered Questions and Future Possibilities
Despite an explosion of cataloged editing sites, scientists still struggle to separate meaningful edits from background noise. Many detected changes may have little impact, but a minority clearly have life-or-death consequences for cells and organisms. The authors argue that future work must identify which enzymes and helper proteins control individual sites, and then test what happens when those single bases are forced to be always edited or never edited. Such studies will clarify how RNA editing contributes to specific illnesses and reveal whether adjusting editing patterns could become a new class of precision therapies—for calming an overactive immune system, tuning brain circuits, correcting metabolic imbalances or making cancers more vulnerable to treatment.
Why This Matters for Everyday Health
In simple terms, this article shows that our cells do not just read the genetic code; they actively proofread and revise it at the RNA stage, using two parallel "editing codes". When these tiny edits occur in the right place and amount, they help keep our immune system balanced, our brains stable, our metabolism flexible and our defenses against viruses sharp. When the editing machinery is overactive, misplaced or broken, those same changes can tip us toward autoimmunity, infection, dementia, metabolic disease or cancer. By mapping and understanding these single-letter rewrites, scientists hope to diagnose disease earlier, predict who is at risk, and eventually design treatments that nudge the editing process back toward health.
Citation: Min, D.J., Lee, S., Lee, Ys. et al. Two codes of RNA editing by deamination in human diseases. Exp Mol Med 58, 382–395 (2026). https://doi.org/10.1038/s12276-025-01633-8
Keywords: RNA editing, ADAR, APOBEC, autoimmunity, cancer